Research Article: Journal of Drug and Alcohol Research (2026) Volume 15, Issue 3
Neurolupus and Updated Therapeutic Drug
Lourdes de Fatima Ibanez Valdes1 and Humberto Foyaca Sibat2*2Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa, South Africa
Humberto Foyaca Sibat, Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa, South Africa, Email: humbertofoyacasibat@gmail.com
Received: 22-Sep-2025, Manuscript No. JDAR-26-171191; Editor assigned: 25-Sep-2025, Pre QC No. JDAR-26-171191 (PQ); Reviewed: 09-Oct-2025, QC No. JDAR-26-171191; Revised: 03-Mar-2026, Manuscript No. JDAR-26-171191 (R); Published: 10-Mar-2026, DOI: 10.4303/JDAR/236500
Abstract
Introduction: Systemic Lupus Erythematosus (SLE) is an autoimmune disease that affects many organs and systems, including the kidneys, neurological system, blood cells, skin, joints, and serous membranes. It is characterized by a broad, varied clinical phenotype and course. Goals: To look for new knowledge.
Methods: To discover articles pertaining to new information on GA and diagnostic techniques, a thorough search of the medical literature was conducted using the databases PubMed/MEDLINE, Scopus, and Embase. We used the PRISMA standards to search the medical literature from January 1, 1990, to January 30, 2026. The following terms were used by the authors to search the scientific databases Scopus, Embassy, Medline, and PubMed Central: “systemic lupus erythematosus” OR “neurorolupus” OR “neuropsychiatric systemic lupus erythematosus” OR “lupus cerebritis” OR “autoantibody” OR “ “neurological symptoms of SLE” or “CNS lupus.”
Results: 297 articles were found through a literature search. 108 publications were chosen after 189 duplicate titles and abstracts were eliminated. 35 research examined the impact of autoantibodies on the pathophysiology of NPSLE after 53 papers were eliminated based on the inclusion/exclusion criteria.
Conclusions: Apoptotic debris, INF-alpha production, B cell expression, CD40L, interleukin 21, and helper T cells are the key components in the pathophysiology of NPSLE.
Keywords
Systemic lupus erythematosus; Neuropsychiatric systemic lupus erythematosus; Autoantibodies; Neurological manifestations of SLE; Lupus cerebritis.
Abbreviations
ANA: Anti-Nuclear Antibodies; ACL: Anti-Cardiolipin Antibody; ANuA: Anti-Nucleosome Antibodies; anti-ds DNA: antidouble- stranded DNA antibody; anti-C1q: anti-C1q antibody; anti-Jo-1: anti-histidyl-tRNA synthetase; anti-Ku: antibodies to Ku protein; anti- La: anti-La/SSB antibody; anti-PCNA: anti-Proliferating Cell Nuclear Antigen; anti-PM-Scl: anti-Polymyositis/Scleroderma antibodies; aPLA: antiphospholipid Antibodies; APS: Antiphospholipid Syndrome; anti- RBP: anti-RNA Binding Protein; anti-RNP: anti-Ribonucleoprotein; anti- Ro: anti-Ro/SSA antibody; anti-Sm: anti-Smith antibody; anti-SSA: anti- Sjogren’s Syndrome A; anti-SSB: anti-Sjogren’s Syndrome B; BAFF: B cell-Activating Factor; BILAG: British Isles Lupus Assessment Group; CD: Cluster of Differentiation; CLASI: Cutaneous Lupus Erythematosus Disease Area and Severity Index; CTLA-4: Cytotoxic T-Lymphocyte- Associated protein 4; ELISA: Enzyme-Linked Immunosorbent Assays; ENA: Extractable Nuclear Antigen; HR: Hazard Ratio; ICOS: Inducible T-cell Costimulator; IFN: Type I Interferon, IFNAR: Interferon-Alpha Receptor; IIFA: Indirect Immunofluorescence Assay; IL-21: Interleukin-21; JAKs: Janus Kinases; LLADAS: Lupus Low Disease Activity State; MMF: Mycophenolate Mofetil; NET: Neutrophil Extracellular Trap; pDCs: plasmacytoid Dendritic Cells; SELENA-SLEDAI: Safety of Estrogens in Lupus Erythematosus National Assessment-Systemic Lupus Erythematosus Disease Activity Index; SLE: Systemic Lupus Erythematosus; STAT: Signal Transducer and Activator of Transcription; TLR: Toll-Like Receptors
Introduction
Large immunological dysregulation and the development of Autoantibodies (AA) that target cytoplasmic, nuclear, and cell surface antigens are hallmarks of Systemic Lupus Erythematosus (SLE), a well-known chronic autoimmune disease. These AA constitute the primary problem in the pathophysiology of SLE, causing organ damage, immune complex formation, and important prognostic and diagnostic indicators. In addition to guiding clinical decision-making and providing more insight into the mechanism of illness manifestation and therapy response, the detection of AA serves to validate and diagnose SLE. Anti-dsDNA, Anti-Nuclear Antibodies (ANA), and anti- Sm antibodies are examples of traditional markers that are still used as diagnostic benchmarks. However, because of their correlations with certain illness morphologies and expression, Anti-Nucleosome Antibodies (ANuA), anti-C1q, anti-ribosomal P, anti-cytokine antibodies, and antiphospholipid receive more progressive scientific interest. Additionally, the previously mentioned AA can indicate the expression of the disease, identify a particular organ that is impacted, or forecast flare-ups. The mechanisms that underlie their persistence include long-lived plasma cell expression and B cell tolerance failure. In conclusion, SLE affects a variety of organs and systems, including the kidneys, neurological system, blood cells, skin, joints, and serous membranes [1]. According to some writers, SLS has a 9:1 female-to-male ratio and is more common in females around childbearing age [2]. The majority of SLE cases are found in individuals with Asian, African, and Hispanic ancestry. While many patients come from locations with little access to healthcare, other ethnic and socioeconomic factors exhibit completely different patterns [3].
By attacking cytoplasmic, nuclear, and cell-surface antigens, several broad-spectrum autoantibodies are crucial to the pathophysiology of SLE [4], resulting in the development of immune complexes, tissue inflammation, and complement activation [5]. Therefore, it is essential to find the particular autoantibodies included in the 2019 EULAR/ACR classification criteria in order to establish the diagnosis of SLE [6]. It has been demonstrated by other writers that some autoantibodies are linked to the clinical signs of SLE [7]. To put it another way, SLE is a long-term autoimmune condition marked by cytoplasmic involvement, extensive immunological dysregulation, and the generation of autoantibodies that target nuclear and cellsurface antigens. In addition to being crucial diagnostic and prognostic indicators, these autoantibodies play a major role in the development of immune complexes, organ damage, and disease pathogenesis [8]. About 20–40% of lupus patients have brain damage due to Systemic Lupus Erythematosus (SLE) [9]. Women in their second to fifth decades of life are most likely to experience it [10,11]. Neurologic consequences of SLE may be linked to vascular illness or localized diseases of uncertain aetiology [12]. A 42-year-old woman with a mixed connective tissue disease Systemic Lupus Erythematosus (SLE), Systemic Sclerosis (SSc), and acute renal failure presented with Raynaud’s phenomenon (with complex digital ulcers), inflammatory arthralgia, synovitis, muscle pain, telangiectasia, severe neurological symptoms (confusion, obnubilation-neurolupus), malignant hypertension, PRES, and esophagitis linked to anti-Sm antibodies.
However, a progressive lupus mouse model can replicate many of the organ and serologic signs of SLE, such as enhanced lymphocyte-driven pathology in the skin, kidneys, and brain, as well as anti-dsDNA antibodies [13,14].
According to Pagkopoulou et al. conventional indicators such anti-Sm antibodies, anti-dsDNA antibodies, and Antinuclear Antibodies (ANA) are present in SLE cases. However, a new confirmatory set of antibodies, including Anti-Nucleosome Antibodies (ANuA), anti-C1q, antiribosomal P, antiphospholipid, and anti-cytokine antibodies, was introduced as a diagnostic cornerstone for SLE due to their ability to predict flares and disease activity under long-lived plasma cell expression and B cell tolerance failure, and to be linked to specific disease phenotypes and organ involvement [8].
Despite the diagnostic difficulty, certain clinical manifestations and specific autoantibody positivity must be recorded in order to validate the diagnosis of SLE since clinical characteristics are not all of them can be seen at once, and they are often ambiguous and subtle in beginning. We postulated that the most popular laboratory test for SLE screening, antinuclear antibody, may be nonspecific and can detect a wide range of different illnesses, even in healthy individuals. Nevertheless, the present classification SLE criteria, such the SLICC [15] or EULAR, may be useful in directing its application in evaluating lab results and symptoms. However, it is crucial to remember that these are not diagnostic standards.
Saffioti et al. documented an unusual case of Whipple illness in 2024 that involved isolated transverse myelitis in the spinal cord and was treated with immunosuppressive medication for SLE [16,17].
DNA replication and repair depend heavily on the activity of the 3′–5′ DNA exonuclease.
It helps fix mistakes made during nucleotide incorporation by enabling DNA polymerases to extract nucleotides from the 3′ end of the developing DNA strand. However, Rioux and colleagues’ meta-analysis of published research revealed no evidence linking SLE to TREX1 risk variations [18].
This review’s primary goals are to provide evidence on the relationship between antibodies and targeted organs, highlight the importance of important AA, and synthesize the most recent information on the pharmacological treatment of SLE.
Materials and Methods
To discover articles pertaining to new information on GA and diagnostic techniques, a thorough search of the medical literature was conducted using the databases PubMed/ MEDLINE, Scopus, and Embase.
We used the PRISMA standards to search the medical literature from January 1, 1990, to January 30, 2026. The terms “systemic lupus erythematosus” OR “ neurolupus” OR “neuropsychiatric systemic lupus erythematosus” OR “lulus cerebritis” OR “autoantibody” OR “CNS lupus” OR “neurological manifestations of SLE” were used by the authors to search the scientific databases Scopus, Embassy, Medline, and PubMed Central.
This study’s systematic review adhered to PRISMA’s suggested guidelines (2020 statement).
Search strategy
The databases PubMed Central/MEDLINE, Scopus, and Embase were thoroughly searched in order to find publications pertaining to new information about the pathophysiology of GA, treatment, and diagnostic techniques. Only articles written in Portuguese, Spanish, and English were chosen. Preclinical research, conference proceedings, and editorials were not included.
Selection of study
To ascertain if the chosen publications were eligible for inclusion, both investigators (LdeFIV and HFS) independently examined the full-text versions of the identified articles as well as the abstracts and titles of those publications. For this systematic analysis, medical studies that provided information on the diagnostic process to evaluate GA were chosen. Additionally, publications that lacked a defined process for validating diagnosis, lacked analysis, had insufficient data, or did not specify the precise number of patients were excluded from consideration.
Selection criteria
Among the requirements were the following: Articles that go into great detail into drug management and/ or pathophysiology. LIS clinical characteristics and demographic data.
The following criteria were used for exclusion:
• Full text inaccessibility.
• Articles with unclear pathogenesis.
• Lack of pertinent clinicopathological data
Non-original studies (e.g., editorials, letters, conference proceedings, book chapters).
• Animal model studies.
• Non-Spanish/Portuguese/English studies.
The papers that were not evaluated completely were eliminated.
Data extraction and quality assessment
According to the National Institutes of Health guidelines and the Quality Assessment of Diagnostic Accuracy Studies version 2 (QUADAS-2) evaluation, the studies’ quality assessment, which took into account applicability issues and bias risk, was classified as good, poor, fair, or reasonable. Both writers evaluated the quality independently, and disputes were settled by scientific debate and ultimate consensus.
Data collection, extraction and bias assessment
Two previously referenced writers revised all abstracts and titles that met the inclusion criteria in order to gather pertinent data for the review. Information on the author’s name, age, year of publication, country of information source, study type, radiopharmaceutical used, total number of cases included, and most likely cause of the GA were gathered for every publication chosen for the review. An improved Excel program was used to import the chosen data from qualified publications.
Outcome measures
Our strategy was to choose the most pertinent articles about the practical efficacy and clinical advantages of various GA diagnostic techniques. Data on safety outcomes, such as adverse and significant adverse events, were also found throughout the clinical research.
Statistical analysis
Statistical investigation was done through XLSTAT (addon for Microsoft Excel, version 2021.4.1,
Results and Discussion
Literature search
A literature search turned up 297 articles. 108 publications were chosen after 189 duplicate titles and abstracts were eliminated. 35 research examined the impact of autoantibodies on the pathophysiology of NeL after 53 publications were eliminated based on the inclusion/ exclusion criteria.
The authors employed a QUADAS-2 evaluation to identify low/moderate risk of bias for nearly all publications in order to provide an accurate management of this searching. The current recommended criteria were used to diagnose NeL in all patients. The authors took into account the significant technical variations that resulted from using quite varied diagnostic procedures in multiple investigations.
Notably, small and mixed cohorts with various diagnostic techniques were processed in several papers, which resulted in fewer GA cases investigated using the same protocol.
Brief comments on clinical relevance of key autoantibodies
In addition to their role in disease monitoring, underlying immunopathogenesis, organ involvement, and prognosis, we hypothesized that one of the most significant roles of autoantibodies is to operate as serological biomarkers.
The main identified antibodies in SLE are presented in Table 1.
| Autoantibody | Prevalence | Clinical features | Mechanism of action |
| Anti-dsDNA | 50-70% | Lupus nephritis; flares; complement consumption | Bind double-stranded DNA forming nephritogenic immune complexes, activate complement, and deposit in glomeruli, triggering inflammation and tissue damage. |
| Anti-Sm | 25-30% | Highly specific for SLE; systemic disease; NPSLE | Target snRNPs interfering with RNA splicing; form immune complexes that activate dendritic cells via TLRs, promoting type I IFN production and systemic autoimmunity; contribute to neurotoxicity via CNS penetration. |
| ANA | >95% | Screening; entry criterion in 2019 EULAR/ACR; non-specific | Bind nuclear antigens activate innate immunity via Fc and TLR signaling. |
| Anti-RNP | ~40% | Raynaud’s; arthritis; overlap syndromes | Bind U1-RNP forming immune complexes that activate plasmacytoid dendritic cells via TLR7, enhancing type I IFN production. |
| Anti-La/SSB | 10-15% | Cutaneous lupus; neonatal lupus; hematologic involvement | Bind RNA-associated proteins forming immune complexes that activate Toll-like receptors and type I interferon pathways; mediate immune dysregulation. |
| Anti-Ro/SSA | 30-40% | Cutaneous lupus; photosensitivity; neonatal lupus; hematologic involvement | Target Ro52/Ro60 ribonucleoproteins, forming immune complexes that activate dendritic cells via TLRs; cross placenta; bind cardiac tissue. |
| ANuA | 70-90% | Early SLE; lupus nephritis | Bind nucleosome complexes; promote immune complex formation, activate complement, and mediate glomerular deposition and glomerular inflammation. |
| Anti-ribosomal P | 10-20% | Psychosis; depression; neuropsychiatric lupus | Target ribosomal P proteins; potential CNS penetration, disrupt neuronal function, and trigger neuroinflammation via cytokine release and immune complex formation. |
| Anti-C1q | 15-45% | Lupus nephritis | Bind the collagen-like region of C1q, impair apoptotic cell clearance, activate complement, and promote immune complex-mediated inflammation. |
| Anti-histone | ~30% | Drug-induced lupus | Bind to histone proteins within chromatin forming immune complexes; activate complement. |
| aPLA (LA, aCL, anti--²2GPI) | 30-40% | Thrombosis; pregnancy loss; anti-phospholipid syndrome | Bind phospholipid- bound proteins; activate endothelial cells, platelets, and complement; activate procoagulants and autoimmune mechanisms. |
| Note: aCL: anti-cardiolipin antibody; ANA: Antinuclear Antibody; ANuA: Anti-Nucleosome Antibody; Anti-C1q: Anti-C1q Complement antibody; Anti-dsDNA: Anti-double stranded DNA antibody; Anti-La/SSB: Anti-La/Sj gren’s Syndrome Type B antibody; Anti-histone: Anti-histone antibody; aPLA: anti-Phospholipid Antibodies; Anti-RNP: Anti-Ribonucleoprotein Antibody; Anti Ro/SSA: anti-Ro/Sj¶gren’s Syndrome Type A antibody; Anti-Sm: Anti-Smith antibody; Anti-ribosomal P: anti-Ribosomal P protein antibody; anti-²2GPI: anti-²2 glycoprotein I antibody; LA: Lupus Anticoagulant; snRNPs: small nuclear Ribonucleoproteins. | |||
Table 1: Key SLE autoantibodies, prevalence, features, and mechanisms summarized
Antinuclear Antibodies (ANA) are the most sensitive and specific in over 95% of SLE patients, and they can be detected before clinical symptoms appear in a significant number of SLE cases [19,20]. However, some connective tissue illnesses and even some healthy people can have lower levels of ANA positive. However, the 2029 EULAR/ ACR categorization scheme (titer ≥ 1:80) requires ANA positive for admission. However, ANA in SLE can be generically classified as targeting DNA or RNA-associated protein complexes, anti-Extractable Nuclear Antigen (ENA) antibodies, and anti-DNA/nucleosome antibodies [21]. However, anti-double-stranded DNA (anti-dsDNA) antibodies (antibodies against double-stranded DNA) are very specific for diagnosis in 50–70% of SLE patients [22]. Other autoantibodies: According to some authors, 50% of SLE patients have anti-RNA Binding Protein (anti-RBP) autoantibodies. These autoantibodies come in a variety of subtypes, including anti-Ribonucleoprotein (anti-RNP), anti-Ro/SSA, anti-Smith (anti-Sm), and anti-La/SSB, all of which target particular RNA-protein complexes and are associated with particular clinical characteristics [23]. With the exception of SLE neuropsychiatric disorders (acute confusional states), some of them, such as anti- Sm antibodies, have high specificity but poor sensitivity (<20%) and are only found in a small number of cases (25–30%) specifically directed against small nuclear ribonucleoproteins, primarily in Asian and African peoples, with no strong correlation to clinical features but exhibiting elevated diagnostic specificity [24–26].
While their presence indicates widespread B cell activation and overlapping autoimmunity, some products of longlived plasma cells, such as anti-RNP antibodies, can react to persistent infections or vaccinations but are not indicators of illness flare-ups in SLE [27]. Thirty to forty percent of SLE patients have Anti-Sjogren’s syndrome A (anti/SSAAnti SSAAnti- Ro), and ten to fifteen percent have Anti-Sjogren’s syndrome B (anti-SSB/Anti La) [28].
Maternal anti-Ro antibodies are notably associated with congenital heart block and neonatal lupus, whereas the aforementioned autoantibodies are frequently found in cases exhibiting photosensitivity, skin manifestations, vasculitis (palpable purpura), thrombocytopenia, anemia, and leukopenia [29,30]. These antibodies’ importance in early illness pathogenesis is highlighted by the fact that they can be detected years before SLE clinical signs appear [31]. When Antiphospholipid Syndrome (APS) is linked to SLE, its immunological background is supported by the existence of antiphospholipid Antibodies (aPLA), such as anti-cardiolipin, lupus anticoagulant, and anti-β2 glycoprotein I antibodies [32]. Venous and arterial thrombosis, thrombocytopenia, anemia, pre-eclampsia, livedo reticularis, and neurologic symptoms including migraine or cognitive impairment are all associated with APS [33-36].
Targeting ribosomal P proteins, anti-ribosomal P antibodies have been linked (10 and 47%) to neuropsychiatric SLE. Ethnicity, geography, immunoassay type, cohort design, and age at disease beginning are some of the variables that affect it [37].
Since ANuA is positive in 70-90% of patients, it may encourage immune complex development and deposition on glomerular basement membranes, starting inflammatory cascades.
Anti-dsDNA antibody development may be preceded by renal involvement, elevated disease activity, and enhanced capillary permeability [38,39].
Conversely, approximately 30% of people with idiopathic lupus and over 75% of people with drug-induced SLE (isoniazid and penicillamine) have anti-histone antibodies [40,41].
Anti-C1q antibodies, a novel biomarker for lupus nephritis that is directly linked to renal flare-ups and histological activity, have surfaced in 15–45% of SLE cases and have predictive value on complement levels and in conjunction with anti-dsDNA. Additionally, it has a notable impact on the apoptotic cell clearance mechanism [42]. Other autoantibodies, such as anti- Proliferating Cell Nuclear Antigen (anti-PCNA) antibodies, which are relatively rare with a prevalence of 1–10% and frequently linked to hepatic involvement and active SLE, have been confirmed in lupus patients in addition to the previously mentioned ones [43]. Antibodies to anti-Polymyositis/ Scleroderma antibodies (anti-PM-Scl) and Ku protein (anti- Ku) are commonly observed, and other comorbidities with SLE, such as systemic sclerosis or polymyositis, may indicate the presence of mixed connective tissue phenotype [44,45].
While anti-centromere and anti-Scl-70 (topoisomerase I) antibodies are frequently seen in cases of systemic sclerosis, they can also occasionally be seen in SLE patients with sclerodermatous symptoms or Raynaud’s syndrome [46]. Anti-synthetase syndrome includes certain anti-synthetase antibodies, such as anti-histidyl-tRNA synthetase (anti- Jo-1). However, proximal muscular weakness, interstitial lung disease, or even myositis may be accompanied by these antibodies [47]. Anti-fibrillarin antibodies and Mi-2 beta antigen (anti-Mi-2) may be found in patients with polymyositis/dermatomyositis and systemic sclerosis [48]. Autoantibodies that target cytokines like IP-10 [49], INF I-II [50], and BAFF [51] have been shown to be one of the factors that predict disease manifestation and flare-ups in SLE patients.
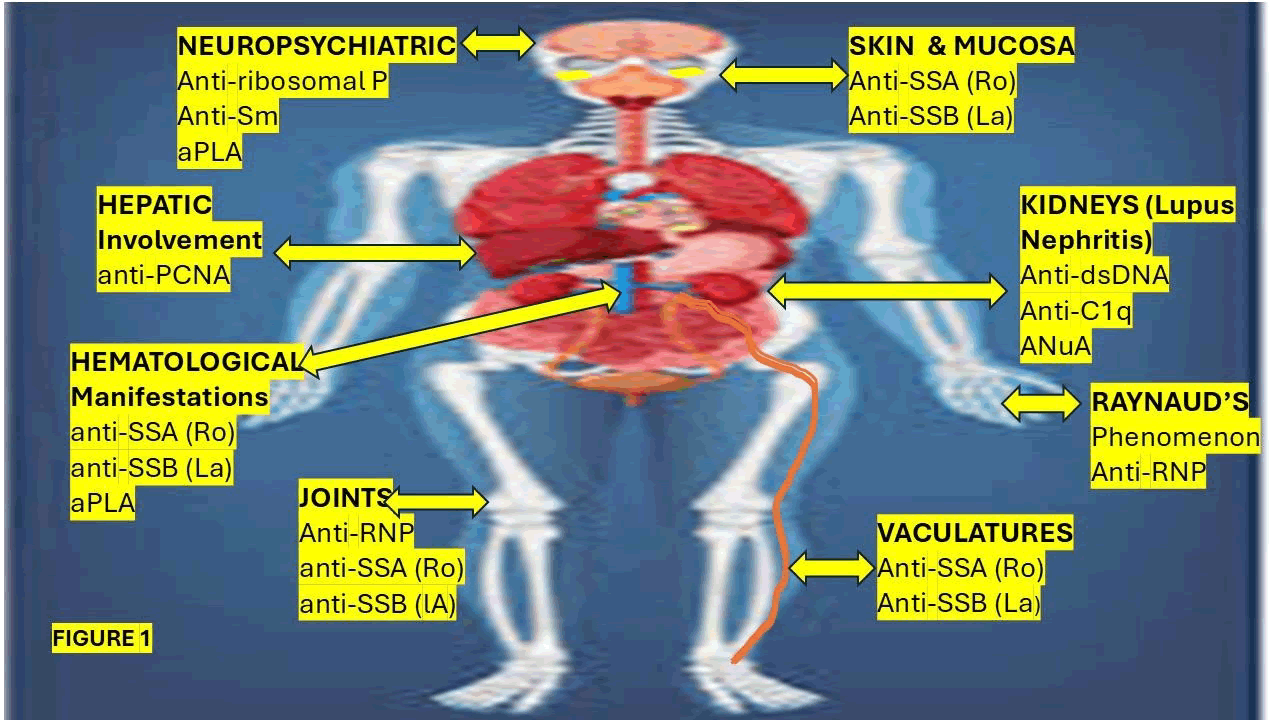
The main antibodies SLE-targeting different organs and their corresponding autoantibody association are represented in Figure 1.
Figure 1: Shows graphical representation of the major SLE-related affected organs and their corresponding autoantibody associations
Brief comments on drugs implications and therapeutic emerging targets
After administering rituximab to 190 patients, Silva Brito et al. came to the conclusion that the drug is highly beneficial in SLE cases, primarily in those with refractory or lifethreatening neuropsychiatric or nephritic SLE signs [52].
In order to control antibody-mediated inflammation, immunosuppressive medications like glucocorticosteroids, azathioprine, hydroxychloroquine, mycophenolate mofetil, and cyclophosphamide should be prescribed based on the pathophysiology and progression of SLE [53].
Using anti-BAFF drugs, type I IFN receptor blockage, anti-CD20 monoclonal antibodies, and IFN signaling suppression, the therapy program’s primary goal is to reduce the formation of harmful autoantibodies [54].
The new and emerging drugs targeting autoantibodies in SLE are summarized in Table 2.
| Medication | Target | Pharmacology | Indication | Clinical results |
| Belimumab | BAFF inhibitio n | Inhibits B cell survival | Non-renal and renal SLE | Improved SRI-4 and renal response in BLISS-52/76 and BLISS-LN trials; FDA-approved; steroid-sparing. Significant improvement in refractory SLE and efficacy in pediatric SLE. |
| Rituximab | CD20+ B cells | Depletes mature B cells; reduces autoantibody production | Refractory SLE/LN/NPSLE | Similar to placebo effects in the LUNAR trials. Off-label use supported in refractory NPSLE (85% response, 45% relapse), reduces anti-dsDNA, steroid-sparing. |
| Obinutuzu mab | CD20+ B cells | Type II anti-CD20 antibody; induces enhanced B cell apoptosis | Refractory LN post-rituximab | NOBILITY:improved CRR (41% vs. 23%, p=0.026) with improvements from baseline in C3, C4 anti-dsDNA and eGFR (adjusted mean difference, 9.7 mL/min/1.73 m2 (95% CI 1.7–18), p=0.017). |
| Anifrolum a b | IFNAR1 | Blocks type I IFN receptor, inhibiting IFN signaling | Non-renal SLE | MUSE: increased SRI-4 (34.3% vs. 17.6%, p=0.014), benefit in IFN-high. TULIP-1: no SRI-4 benefit, signal in BICLA and CLASI. TULIP-2: ↑BICLA (47.8% vs. 31.5%, p=0.001), steroid- sparing, skin benefits. |
| Sifalimuma b | IFN-α | Neutralizes most IFN-α subtypes | SLE | Phase IIb: Improved SRI-4 at week 52 (58–60% vs. 45%), broad disease activity improvement, increased occurrence of herpes zoster infection. |
| Dapirolizu mab | PEGylat ed anti-CD40L Fab | Inhibits T cell–B cell interaction via CD40–CD40L axis | SLE | Phase II: modest improvement in BICLA, SRI-4, and serologic markers vs. placebo; dose– response not met (p=0.07). |
| Abatacept | CD80/CD86 | Inhibits CD28 co-stimulation on T cells | SLE, LN | ACCESS: no improvement in CRR at 24 weeks (33% vs. 31%); 50% of abatacept responders sustained remission after stopping immunosuppress sants. |
| Tofacitinib | JAK 1/3 | Modulates type I IFN responses and T cell activation | Investigational | Phase I: improved HDL profile, vascular function, and IFN signature in SLE; benefits stronger in STAT4-risk carriers. |
| Baricitinib | JAK 1/2 | Inhibits JAK-STAT signaling; reduces inflammator y cytokine signaling | Cutaneous/articular SLE | Improvement in arthritis/rash resolution at week 24 (67% vs. 53%, p=0.041); preclinical data support renoprotection via JAK/STAT modulation. |
| Note: BICLA: BILAG-Based Composite Lupus Assessment; CD: Cluster of Differentiation; CI: Confidence Interval; CLASI: Cutaneous Lupus Erythematosus Disease Area and Severity Index; CRR: Complete Renal Response; dsDNA: double-stranded Deoxyribonucleic Acid; eGFR: estimated Glomerular Filtration Rate; FDA: Food and Drug Administration; HDL: High-Density Lipoprotein; HR: Hazard Ratio; IFN: Interferon; IFNAR: Interferon-α/β Receptor; ISG: Interferon-Stimulated Genes; ISM: Interferon Signature Metric; JAK: Janus Kinase; LN: Lupus Nephritis; NPSLE: Neuropsychiatric Systemic Lupus Erythematosus; PEG: Polyethylene Glycol; SLE: Systemic Lupus Erythematosus; SRI-4: SLE Responder Index (≥ 4 point improvement); STAT: Signal Transducer and Activator of Transcription. | ||||
Table 2: Targeted therapies in SLE against specific autoantibodies or their production.
Rituximab, a chimeric monoclonal antibody that targets CD20 and depletes circulating B lymphocytes, is the most commonly prescribed treatment medication in patients with refractory SLE, primarily those with neuropsychiatric indications of lupus nephritis [55,56].
However, rituximab has not improved outcomes for African American and Hispanic patients [57].
In a different study, 35 patients with refractory NeL responded to Rituximab with an 85% response rate [58].
There are currently no large-scale randomized controlled clinical trials on rituximab in SLE. Belimumab, a monoclonal antibody that targets BAFF to reduce the survival of autoreactive B cells and lower disease expression and flareups in SLE patients, was the first biological medication to treat SLE. In over 40% of instances, belimumab at a dose of 10 mg/kg has demonstrated good efficacy [59].
Some researchers found that 58% of patients had positive outcomes [60], while another found benefits of up to 70% [61].
Anifrolumab, an IgG1k monoclonal antibody that can bind to subunit 1 of the type-1 IFN receptor (IFNAR) and inhibit the formation of the IFN/IFNAR complex and subsequent gene transcription, was reported by Peng and colleagues as an anti-interferon-α receptor 1 antibody with significant advantages over placebo in SLE cases at a dose of 300 mg/4 weeks [62].
However, other researchers found increases in quality of life scores, stable organ damage, serologic improvement, and acceptable safety of 69.7% [63].
Being the second preferred medication that the FDA has licensed to treat SLE patients who do not have nephritis lupus [64]. In addition to rituximab, obinutuzumab is another anti-CD20 monoclonal antibody that can enhance Fc effector activity and cause B cell death at a dose of 200 mg. Consequently, it is recommended for the treatment of B cell cancers [65].
It has demonstrated remarkable efficacy in clinical efficacy, lupus nephritis remission, B cell depletion, decreased anti- RNA antibodies, and CD4+ T cell activation in animal studies; in humans, it has improved C3 and anti-dsDNA levels and allowed the tapering of corticosteroids compared to rituximab in the majority of patients [66,67].
The efficacy of rontalizumab, an anti-IFN-α monoclonal antibody with the ability to neutralize all 12 IFN-α subtypes, in patients with moderate-to-severe SLE was evaluated in a clinical trial in which it was able to simultaneously lower steroid consumption, increase SRI responses, and lower flare rates when compared to a placebo [68].
However, sifalimumab, another human IgG1κ antibody, is a well-tolerated medication for SLE patients and can neutralize the majority of IFN-α subtypes. Other researchers demonstrated its effectiveness with notable improvements in herpes zoster infections and a higher SRI response rate at week 52 compared to placebo (58–60% vs. 45%) [69].
Abatacept, a fusion protein, has been developed as a treatment option for SLE and licensed for rheumatoid arthritis in order to target T cell co-stimulation pathways. It modifies CD80/CD86:CD28 signaling by combining the Fc region of IgG1 with cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) [70].
Lastly, several authors suggested that the Janus Kinases (JAKs) and Signal Transducer and Activator Of Transcription (STAT) pathways are essential components of immunological tolerance, and that their dysregulation causes autoimmune disorders like SLE [71].
Based on its safety and improvement in cardiometabolic and immunologic markers, including type I IFN signature, another JAK 1/3 inhibitor, such as tofacitinib, has been suggested for pharmacological therapy of SLE [72].
Conclusion
In conclusion, we came to the conclusion that AA is crucial for verifying the final diagnosis, understanding the pathophysiology of the condition, and establishing SLE stratification that sheds light on the disease’s heterogeneity and prognosis. Based on our analysis of the medical literature, we emphasized that anti-dsDNA and anti-C1q antibodies maintain a remarkable predictive value in lupus nephritis, while the confirmation of anti-Sm, anti-Ro, and anti-ribosomal P antibodies supports the diagnosis of hematologic phenotypes, cutaneous manifestations of SLE, or neuropsychiatric lupus. In order to better understand the mechanisms behind long-term outcomes in SLE, we predicted that biomarker validation would be essential. Following a thorough analysis of the available medical literature, we came to the conclusion that the most effective medication therapeutic management of Nel greatly depends on the underlying cause of the active NP manifestation, immunosuppressive pharmacology for inflammatory and autoimmune manifestations, anti-thrombotic therapy for vascular disorders, and symptomatic treatment. Unfortunately, there are very few clinical trial studies pertaining to NeL medication therapy. Thus, future research on therapeutic medication management for NeL should be carried out in order to add to the body of knowledge capable of offering a long-term curative treatment for NeL.
Acknowledgment
To thanks to Prof Thozama Dubula for his unconditional support.
Ethics Statement
This review does not require ethical approval.
Patient Privacy
All patientâidentifying information has been removed to ensure anonymity.
Conflicts of Interest
Authors of this review report there is not conflicts of interest.
References
- G.C. Tsokos, M.S. Lo, P.C. Reis, K.E. Sullivan, New insights into the immunopathogenesis of systemic lupus erythematosus, Nat Rev Rheumatol, 12(2016):716–730.
[Crossref] [Google Scholar] [PubMed]
- A. Rahman, D.A. Isenberg, Systemic lupus erythematosus, N Engl J Med, 358(2008):929–939.
[Crossref] [Google Scholar] [PubMed]
- M.R.W. Barber, C. Drenkard, T. Falasinnu, A. Hoi, A. Mak, et al. Global epidemiology of systemic lupus erythematosus, Nat Rev Rheumatol, 17(2021):515–532.
[Crossref] [Google Scholar] [PubMed]
- B. Dema, N. Charles, Autoantibodies in SLE: Specificities, isotypes and receptors, Antibodies, 5(2016):2.
[Crossref] [Google Scholar] [PubMed]
- V.M. Holers, Systemic lupus erythematosus as the paradigm for understanding complex immune relationships and complement targeting, Immunobiology, 230(2025):152915.
[Crossref] [Google Scholar] [PubMed]
- M. Aringer, K. Costenbader, D. Daikh, R. Brinks, M. Mosca, et al. 2019 EULAR/ACR classification criteria for SLE, Arthritis Rheumatol, 71(2019):1400–1412.
[Crossref] [Google Scholar] [PubMed]
- M. Abraham, C.T. Derk, Anti-ribosomal-P antibodies in lupus and related conditions, Rheumatol Int, 35(2015):27–33.
- E. Pagkopoulou, C. Loutradis, M. Papaioannou, M. Daoudaki, M. Stangou, et al. Autoantibodies in systemic lupus erythematosus: Diagnostic and pathogenic insights, J Clin Med, 14(2025):5714.
[Crossref] [Google Scholar] [PubMed]
- S. Kivity, N. Agmon-Levin, G. Zandman-Goddard, J. Chapman, Y. Shoenfeld, Neuropsychiatric lupus: A mosaic of clinical presentations, BMC Med, 13(2015):43.
[Crossref] [Google Scholar] [PubMed]
- J.S. Nusbaum, I. Mirza, J. Shum, R.W. Freilich, R.E. Cohen, et al. Sex differences in systemic lupus erythematosus, Mayo Clin Proc, 95(2020):384–394.
- E.C. Somers, W. Marder, P. Cagnoli, E.E. Lewis, P. DeGuire, et al. Population-based incidence and prevalence of SLE, Arthritis Rheumatol, 66(2014):369–378.
- J. Reynolds, M. Huang, Y. Li, M. Meineck, T. Moeckel, et al. IL-6 knockout ameliorates neuropsychiatric lupus features, J Neuroinflammation, 21(2024):89.
- C. Gros, O. Fogel, I. Boudhabhay, C. Debiais, J.P. Duong Van Huyen, et al. Renal failure with neurological symptoms in mixed connective tissue disease, J Scleroderma Relat Disord, 8(2022):NP6–NP10.
[Crossref] [Google Scholar] [PubMed]
- D. Perry, A. Sang, Y. Yin, Y.Y. Zheng, L. Morel, Murine models of systemic lupus erythematosus, J Biomed Biotechnol, 2011(2011):271694.
[Crossref] [Google Scholar] [PubMed]
- M. Petri, A.M. Orbai, G.S. Alarcón, C. Gordon, J.T. Merrill, et al. SLICC classification criteria for SLE, Arthritis Rheum, 64(2012):2677–2686.
- M. Aringer, K. Costenbader, D. Daikh, R. Brinks, M. Mosca, et al. 2019 EULAR/ACR classification criteria for SLE, Ann Rheum Dis, 78(2019):1151–1159.
- C. Saffioti, M. Nebiolo, R. Caorsi, A. Mesini, M. Severino, et al. Whipple disease in SLE: Case report and review, Infect Dis Rep, 16(2024):269–280.
- B. Rioux, S. McGlasson, D. Forbes, K.R. Reid, A. Klingseisen, et al. Type I interferon signatures and SLE risk, Nat Commun, 17(2026):1073.
- M. Mahler, P.L. Meroni, X. Bossuyt, M.J. Fritzler, Autoantibodies to cellular antigens (ANA), J Immunol Res, 2014(2014):315179.
- D.S. Pisetsky, Antinuclear antibody testing—misunderstood or misbegotten?, Nat Rev Rheumatol, 13(2017):495–502.
[Crossref] [Google Scholar] [PubMed]
- K.H. Ching, P.D. Burbelo, C. Tipton, C. Wei, M. Petri, et al. Two major autoantibody clusters in SLE, PLoS One, 7(2012):e32001.
[Crossref] [Google Scholar] [PubMed]
- A. Rahman, Autoantibodies, lupus and the science of sabotage, Rheumatology, 43(2004):1326–1336.
[Crossref] [Google Scholar] [PubMed]
- F. Blanco, J. Kalsi, D.A. Isenberg, Antibodies to RNA in SLE and related diseases, Clin Exp Immunol, 86(1991):66–70.
[Crossref] [Google Scholar] [PubMed]
- E.J. ter Borg, G. Horst, P.C. Limburg, C.G. Kallenberg, Anti-Sm antibodies in SLE, J Autoimmun, 4(1991):155–164.
- E. Cozzani, M. Drosera, G. Gasparini, A. Parodi, Serology of lupus erythematosus, Autoimmune Dis, 2014(2014):321359.
- S. Hirohata, Y. Sakuma, T. Yanagida, T. Yoshio, CSF anti-Sm antibodies and neuropsychiatric SLE, Arthritis Res Ther, 16(2014):450.
- D.S. Pisetsky, Evolving story of autoantibodies in SLE, J Autoimmun, 110(2020):102356.
[Crossref] [Google Scholar] [PubMed]
- J.G. Routsias, A.G. Tzioufas, B-cell epitopes of Ro/SSA and La/SSB autoantigens, J Autoimmun, 35(2010):256–264.
- B.T. Kurien, J. Newland, C. Paczkowski, K.L. Moore, R.H. Scofield, Neutropenia in SLE associated with anti-Ro antibodies, Clin Exp Immunol, 120(2000):209–217.
- W. Sun, C. Fu, X. Jin, C. Lei, X. Zhu, Neonatal lupus erythematosus: An acquired autoimmune disease, Ann Med, 57(2025):2476049.
[Crossref] [Google Scholar] [PubMed]
- E.K. Chan, J.C. Hamel, J.P. Buyon, E.M. Tan, Molecular definition of SS-A/Ro autoantigen, J Clin Investig, 87(1991):68–76.
- O. Ünlü, S. Zuily, D. Erkan, Clinical significance of antiphospholipid antibodies in SLE, Eur J Rheumatol, 3(2016):75–84.
[Crossref] [Google Scholar] [PubMed]
- J.S. Levine, D.W. Branch, J. Rauch, The antiphospholipid syndrome, N Engl J Med, 346(2002):752–763.
[Crossref] [Google Scholar] [PubMed]
- G. El Hasbani, A.N. Saliba, I. Uthman, A.T. Taher, Hematological manifestations of antiphospholipid syndrome, Blood Rev, 58(2023):101015.
[Crossref] [Google Scholar] [PubMed]
- J.S. Knight, D.W. Branch, T.L. Ortel, Antiphospholipid syndrome: Advances in diagnosis and management, BMJ, 380(2023):e069717.
- M. Leal Rato, M. Bandeira, V.C. Romão, D.A. de Sousa, Neurologic manifestations of antiphospholipid syndrome, Curr Neurol Neurosci Rep, 21(2021):41.
[Crossref] [Google Scholar] [PubMed]
- M.Y. Choi, R.D. FitzPatrick, K. Buhler, M. Mahler, M.J. Fritzler, Anti-ribosomal P autoantibodies in SLE: Meta-analysis, Autoimmun Rev, 19(2020):102463.
[Crossref] [Google Scholar] [PubMed]
- N. Bizzaro, D. Villalta, D. Giavarina, R. Tozzoli, Anti-nucleosome vs. anti-dsDNA antibodies in SLE diagnosis, Autoimmun Rev, 12(2012):97–106.
- O.P. Rekvig, C. Putterman, C. Casu, H.X. Gao, A. Ghirardello, et al. Autoantibodies in lupus: Culprits or bystanders?, Autoimmun Rev, 11(2012):596–603.
[Crossref] [Google Scholar] [PubMed]
- U. Katz, G. Zandman-Goddard, Drug-induced lupus: An update, Autoimmun Rev, 10(2010):46–50.
[Crossref] [Google Scholar] [PubMed]
- X.Y. Sun, J. Shi, L. Han, Y. Su, Z.G. Li, Anti-histone antibodies in SLE, J Clin Lab Anal, 22(2008):271–277.
[Crossref] [Google Scholar] [PubMed]
- M.E. Munroe, J.A. James, Genetics of lupus nephritis, Semin Nephrol, 35(2015):396–409.
- M. Mahler, K. Miyachi, C. Peebles, M.J. Fritzler, Autoantibodies to PCNA: Clinical significance, Autoimmun Rev, 11(2012):771–775.
- Y. Muro, Y. Hosono, K. Sugiura, Y. Ogawa, T. Mimori, et al. Anti-PM/Scl antibodies in autoimmune diseases, Arthritis Res Ther, 17(2015):57.
- K. Alsulami, J. D’Aoust, Mixed connective tissue disease with anti-Ku positivity, Cureus, 16(2024):e72310.
[Crossref] [Google Scholar] [PubMed]
- M. Satoh, E.K. Chan, E.S. Sobel, D.L. Kimpel, Y. Yamasaki, et al. Clinical implication of autoantibodies in rheumatic diseases, Expert Rev Clin Immunol, 3(2007):721–738.
- H. Gunawardena, Z.E. Betteridge, N.J. McHugh, Myositis-specific autoantibodies: Clinical significance, Rheumatology, 48(2009):607–612.
- M.G.P. Cruellas, V.V. dos Santos Trindade, M. Levy-Neto, F.H.C. de Souza, S.K. Shinjo, Myositis autoantibody profiles and clinical associations, Clinics, 68(2013):909–914.
- S. Gupta, I.P. Tatouli, L.B. Rosen, S. Hasni, I. Alevizos, et al. Autoantibodies against interferon in SLE, Arthritis Rheumatol, 68(2016):1677–1687.
- M. Slavikova, H. Schmeisser, E. Kontsekova, F. Mateicka, L. Borecky, et al. Autoantibodies against interferons in SLE patients, J Interferon Cytokine Res, 23(2003):143–147.
- J. Friebus-Kardash, L. Branco, C. Ribi, C. Chizzolini, U. Huynh-Do, et al. BAFF-IgG immune complexes in SLE activity, Nephrol Dial Transplant, 33(2018):54–64.
- A. da Silva Brito, S. Miranda, T. Moitinho de Almeida, D.A. Isenberg, Effect of rituximab on long-term damage in SLE, Rheumatology (Oxford), 64(2025):5031–5036.
- K. Saegusa, Y. Tsuchida, T. Komai, H. Tsuchiya, K. Fujio, Advances in targeted therapy for SLE, Sci, 26(2025 Int J Mol):929.
- V. Venturelli, D.A. Isenberg, Targeted therapy for SLE: Current and future perspectives, J Clin Med, 12(2023):3198.
- K. Arzoo, S. Sadeghi, H.A. Liebman, Rituximab in refractory autoimmune disorders, Ann Rheum Dis, 61(2002):922–924.
- J.T. Merrill, C.M. Neuwelt, D.J. Wallace, J.C. Shanahan, K.M. Latinis, et al. Rituximab efficacy and safety in SLE trial, Arthritis Rheumatol, 62(2010):222–233.
- J. Narváez, V. Ríos-Rodriguez, D. de la Fuente, P. Estrada, L. López-Vives, et al. Rituximab in neuropsychiatric lupus, Semin Arthritis Rheum, 41(2011):364–372.
- R. Furie, M. Petri, O. Zamani, R. Cervera, D.J. Wallace, et al. Belimumab in SLE: Phase III trial, Arthritis Rheumatol, 63(2011):3918–3930.
- S.V. Navarra, R.M. Guzmán, A.E. Gallacher, S. Hall, R.A. Levy, et al. Belimumab efficacy and safety in SLE, Lancet, 377(2011):721–731.
- M. Shipa, A. Embleton-Thirsk, M. Parvaz, L.R. Santos, P. Muller, et al. Ehrenstein, Belimumab after rituximab in SLE, Ann Intern Med, 174(2021):1647–1657.
- R. Furie, M. Khamashta, J.T. Merrill, V.P. Werth, K. Kalunian, et al. Anifrolumab in moderate-to-severe SLE, Arthritis Rheumatol, 69(2017):376–386.
- W.W. Chatham, R. Furie, A. Saxena, P. Brohawn, E. Schwetje, et al. Long-term safety of anifrolumab in SLE, Arthritis Rheumatol, 73(2021):816–825.
- S. Díaz-Planellas, D. Katsifis-Nezis, A. Fanouriakis, Interferon inhibitors in SLE and anifrolumab efficacy, Mediterr J Rheumatol, 35(2024):381–391.
- K. Tobinai, C. Klein, N. Oya, G. Fingerle-Rowson, Obinutuzumab in B-cell malignancies, Adv Ther, 34(2017):324–356.
[Crossref] [Google Scholar] [PubMed]
- A.D. Marinov, H. Wang, S.I. Bastacky, E. van Puijenbroek, T. Schindler, et al. Obinutuzumab vs. rituximab in lupus model, Arthritis Rheumatol, 73(2021):826–836.
- J. Arnold, S. Dass, S. Twigg, C.H. Jones, B. Rhodes, et al. Obinutuzumab in SLE with rituximab non-response, Rheumatology, 61(2022):4905–4909.
- K.C. Kalunian, J.T. Merrill, R. Maciuca, J.M. McBride, M.J. Townsend, et al. W.P. Kennedy, Rontalizumab in SLE (ROSE study), Ann Rheum Dis, 75(2016):196–202.
- M. Khamashta, J.T. Merrill, V.P. Werth, R. Furie, K. Kalunian, et al. Greth, Sifalimumab in SLE, Ann Rheum Dis, 75(2016):1909–1916.
- H.A. Blair, E.D. Deeks, Abatacept in rheumatoid arthritis, Drugs, 77(2017):1221–1233.
[Crossref] [Google Scholar] [PubMed]
- G.S. Moysidou, A. Dara, JAK inhibition in SLE, Mediterr J Rheumatol, 35(2024):37–44.
[Crossref] [Google Scholar] [PubMed]
- S.A. Hasni, S. Gupta, M. Davis, E. Poncio, Y. Temesgen-Oyelakin, et al. Tofacitinib in SLE: Phase 1 trial, Nat Commun, 12(2021):3391.
- G.K. Bertsias, J.P. Ioannidis, M. Aringer, E. Bollen, S. Bombardieri, et al. EULAR recommendations for neuropsychiatric SLE, Ann Rheum Dis, 69(2010):2074–2082.
Copyright: © 2026 Lourdes de Fatima Ibanez Valdes, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.