Research Article: Journal of Drug and Alcohol Research (2022) Volume 11, Issue 2
Validated Rp-Hplc Method Development for Estimation of Cobicistat and Darunavir in Bulk and Dosage Forms
B Rajeswari1*, N Saritha2 and N Devanna12Department of Chemistry, JNTU Engineering College of Kalikiri, India
B Rajeswari, Department of Chemistry, JNTU Anantapur, India, Email: rajeswari@go.id
Received: 26-Jan-2022, Manuscript No. jdar-22-54631;;Accepted Date: Feb 25, 2022; Editor assigned: 28-Jan-2022, Pre QC No. jdar-22-54631 (PQ); Reviewed: 11-Feb-2022, QC No. jdar-22-54631; Revised: 18-Feb-2022, Manuscript No. jdar-22-54631(R); Published: 25-Feb-2022, DOI: 10.4303/jdar/236163
Abstract
A simple, accurate, precise method was developed for the simultaneous estimation of the Cobicistat and Darunavir in Tablet dosage form. Chromatogram was run through BDS (250 mm 4.6 mm, 5 μ). Mobile phase containing Buffer and Acetonitrile in the ratio of 40:50 A:10 M was pumped through column at a flow rate of 1 ml/min. Temperature was maintained at 30°C. Optimized wavelength for Cobicistat and Darunavir was 210 nm. Retention time of Cobicistat and Darunavir were found to be 3.170 min and 3.984 min. %RSD of the Cobicistat and Darunavir were and found to be 1.06 and 1.3 respectively. %Recover was Obtained as 100.71% and 100.2% for Cobicistat and Darunavir. LOD, LOQ values were obtained from regression equations of Cobicistat and Darunavir were 0.107 ppm, 0.326 ppm and 0.333 ppm, 1.009 ppm respectively. Regression equation of Cobicistat is y=10306x+1346, and of Darunavir is y=8883.x+2152.
Introduction
High Performance Liquid Chromatography (HPLC) is the fastest growing analytical technique for analysis of drugs [1-4]. Its simplicity, high specificity and wide range of sensitivity make it ideal for the analysis of many drugs in both dosage forms and biological fluids. Cobicistat is a cytochrome P450 3A (CYP3A) inhibitor that can be used to enhance the pharmacokinetic profile of certain anti-HIV-1 agents. Cobicistat is a monocarboxylic acid amide obtained by formal condensation of the carboxy group of (2S)-2- ({[(2-isopropyl-1,3-thiazol-4-yl)methyl](methyl)carbamoyl} amino)-4-(morpholin-4-yl) butanoic acid with the amino group of 1,3-thiazol-5-ylmethyl [(2R,5R)-5-amino- 1,6-diphenylhexan-2-yl] carbamate [5].
Darunavir is a human immunodeficiency virus type 1 (HIV-1) protease nonpeptidic inhibitor, with activity against HIV. Upon oral administration, darunavir selectively targets and binds to the active site of HIV-1 protease, and inhibits the dimerization and catalytic activity of HIV-1 protease. The IUPAC name of Darunavir is [(3aS,4R,6aR)-2,3,3a,4,5,6a-hexahydrofuro[2,3-b]furan-4- yl] N-[(2S,3R)-4-[(4-aminophenyl)sulfonyl-(2-methylpropyl) amino]-3-hydroxy-1-phenylbutan-2-yl] carbamate as shown in Figures [1-11] [6,7].
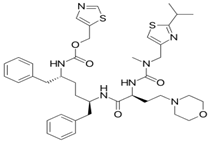
Figure 1: Structure of Cobicistat.
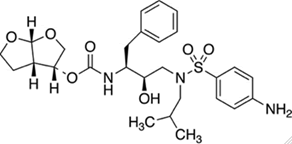
Figure 2: Structure of Darunavir.
Figure 3: Chromatogram of blank.
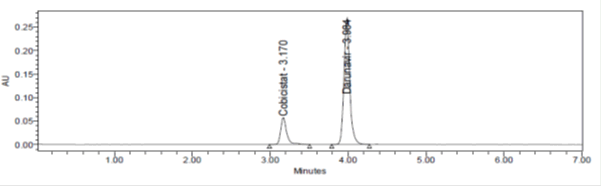
Figure 4: Typical chromatogram of Cobicistat and Darunavir.
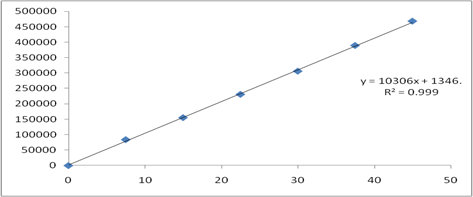
Figure 5: Calibration curve of Cobicistat.
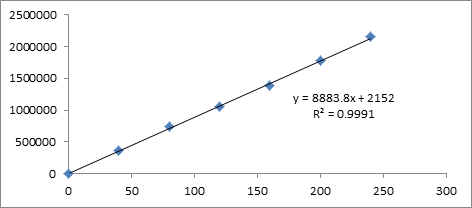
Figure 6: Calibration curve of Darunavir.
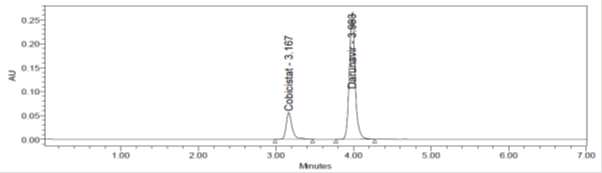
Figure 7: Repeatability Chromatogram of Cobicistat and Darunavir method.
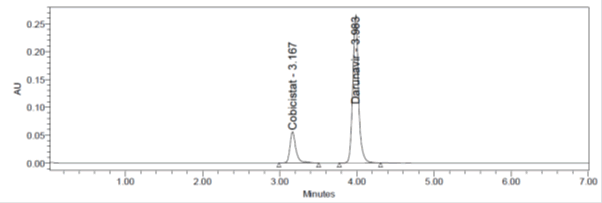
Figure 8: Inter Day precision Chromatogram of Cobicistat and Darunavir method.
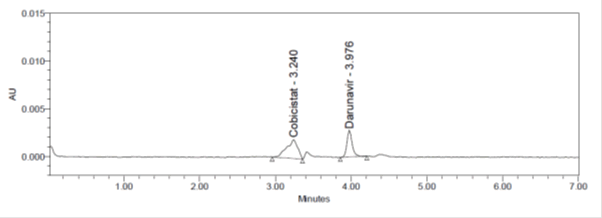
Figure 9: LOD Chromatogram of Cobicistat and Darunavir method.
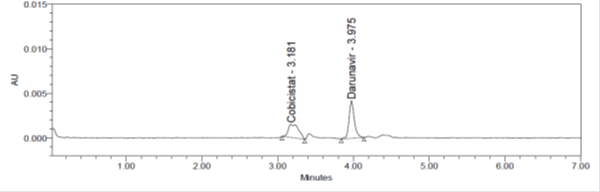
Figure 10: LOQ Chromatogram of of Cobicistat and Darunavir.
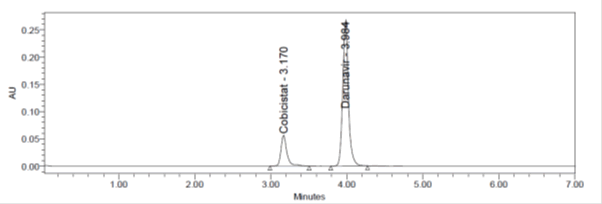
Figure 11: Assay Chromatogram.
Literature review reveals that few analytical methods have been reported for the determination of Cobicistat and Darunavir by using various analytical techniques in individual and in combination with other drugs [8-16]. It was found that no suitable validated method was available from the literature for determination of bioavailability and bioequivalence of Cobicistat and Darunavir in biological samples. The main aim of the present study is to develop a new HPLC method for simultaneous estimation of Cobicistat and Darunavir according to ICH guidelines. To apply a validated technique for the estimation of Cobicistat and Darunavir in pharmaceutical formulation.
Materials and Methods
Cobicistat and Darunavir, Combination of Cobicistat and Darunavir tablet dosage forms, distilled water, acetonitrile, phosphate buffer, ammonium acetate buffer, glacial acitic acid, methanol, potassium dihydrogen phosphate buffer, tetra hydro furan, tri ethyl amine, ortho-phosphoric acid etc.
Buffer: (0.1%OPA):1 ML of Ortho phosphoric acid solution in a 1000 ml of volumetric flask add about 100 ml of milli-Qwater and final volume make up to 1000 ml with milli-Q water
Standard Preparation: Accurately Weighed and transferred working Standards of 7.5 mg of Cobicistat and 40 mg of Darunavir into a 25 ml clean dry volumetric flask, add 3/4th volume of diluent, sonicated for 5 minutes and make up to the final volume with diluents. 1 ml from the above two stock solution was taken into a 10 ml volumetric flask and made up to 10 ml.
Sample preparation
1 tablet was weighed, powdered and then the weight was transferred into a 500 mL volumetric flask, 100 mL of diluent added and sonicated for 25 min, further the volume made up with diluent and filtered. From the filtered solution 1 ml was pipetted out into a 10 ml volumetric flask and made upto 10 ml with diluents.
Linearity
Linearity solutions are prepared such that 0.25, 0.5, 0.75,1, 1.25, 1.5 ml from the Stock solutions of Cobicistat and Darunavir are taken in to 6 different volumetric flasks and diluted to 10 ml with diluents to get 7.5 ppm, 15 ppm, 22.5 ppm, 30 ppm, 37.5 ppm, 45 ppm of Cobicistat and 40 ppm, 80 ppm, 120 ppm, 160 ppm, 200 ppm, 240 ppm of Darunavir.
Accuracy preparations
From the formulation solution take 0.5 ml, 1 ml, 1.5 ml, was transferred to 10 ml volumetric flask and make up the volume to get 50% 100% and 150% solution concentrations.
Method development
Few trials with altering chromatographic conditions were performed for method optimization.
Results and Discussion
System suitability
All the system suitability parameters are within range and satisfactory as per ICH guidelines.
Linearity
Six Linear concentrations of Cobicistat (7.5 ppm-45 ppm) and Darunavir (40 ppm-240 ppm) are prepared and injected. Regression equation of the Cobicistat and Darunavir are found to be, y=10306x+1346, and y=8883.x+2152 and the regression co-efficient was 0.999
Precision
Intraday precision (Repeatability): Intraday Precision was performed and %RSD for Cobicistat and Darunavir were found to be 0.44% and 0.2% respectively.
Inter day precision: Inter day precision was performed with 24 hrs time lag and the %RSD Obtained for Cobicistat and Darunavir were 0.74% and 0.2%.
Accuracy
Three concentrations 50%, 100%, 150%, were injected in a triplicate manner and amount Recovered and % Recovery were displayed in Tables 1-7.
Table 1: System suitability studies of Cobicistat and Darunavir.
| Property | Cobicistat | Darunavir |
|---|---|---|
| Retention time(tR) | 3.170± 0.3min | 3.984±0.3min |
| Theoretical plates(N) | 9443 ± 163.48 | 15131± 163.48 |
| Tailingfactor (T) | 1.29 ± 0.117 | 1.14± 0.117 |
Table 2: Calibration data of Cobicistat and Darunavir.
| S.No | Concentration Cobicistat(µg/ml) | Response | Concentration Darunavir(µg/ml) | Response |
|---|---|---|---|---|
| 1 | 0 | 0 | 0 | 0 |
| 2 | 7.5 | 83972 | 40 | 359687 |
| 3 | 15 | 155266 | 80 | 742168 |
| 4 | 22.5 | 230648 | 120 | 1056971 |
| 5 | 30 | 305803 | 160 | 1384921 |
| 6 | 37.5 | 389008 | 200 | 1774658 |
| 7 | 45 | 467853 | 240 | 2159056 |
Table 3: Repeatability results for Cobicistat and Darunavir.
| Sr. No. | Cobicistat | Darunavir |
|---|---|---|
| 1 | 298971 | 1368391 |
| 2 | 298293 | 1367203 |
| 3 | 300896 | 1372673 |
| 4 | 301407 | 1374295 |
| 5 | 299571 | 1375155 |
| 6 | 301312 | 1370158 |
| Mean | 300075 | 1371313 |
| Std. Dev. | 1313.5288 | 3233 |
| %RSD | 0.44 | 0.2 |
Table 4: Inter day precision results for Cobicistat and Darunavir.
| Sr. No. | Cobicistat | Darunavir |
|---|---|---|
| 1 | 300275 | 1368146 |
| 2 | 303143 | 1373628 |
| 3 | 303350 | 1375389 |
| 4 | 301303 | 1376272 |
| 5 | 302344 | 1370977 |
| Mean | 302861.7 | 1372212 |
| Std. Dev. | 2230.0 | 3402 |
| %RSD | 0.74 | 0.2 |
Table 5: Accuracy results of Cobicistat and Darunavir.
| Sample | Amount added (µg/ml) | Amount Recovered (µg/ml) | Recovery (%) | %RSD |
|---|---|---|---|---|
| Cobicistat | 15 | 15.0195 | 100.13 | 1.06 |
| 30 | 30.045 | 100.15 | 0.63 | |
| 45 | 44.9055 | 99.79 | 0.45 | |
| Darunavir | 80 | 80.17 | 100.21 | 1.14 |
| 160 | 160.53 | 100.33 | 0.71 | |
| 240 | 242.99 | 101.25 | 0.87 |
Table 6: Robustness data of Cobicistat and Darunavir.
| S.NO | Robustness condition | Cobicistat %RSD | Darunavir %RSD |
|---|---|---|---|
| 1 | Flow minus | 0.8 | 1.3 |
| 2 | Flow Plus | 0.2 | 0.4 |
| 3 | Mobile phase minus | 0.2 | 2.5 |
| 4 | Mobile phase Plus | 0.1 | 0.7 |
| 5 | Temperature minus | 0.3 | 2.5 |
| 6 | Temperature Plus | 0.6 | 0.4 |
Table 7: Assay of Tablet.
| S. No. | Cobicistat %Assay | Darunavir %Assay |
| 1 | 101.57 | 99.79 |
| 2 | 100.53 | 98.15 |
| 3 | 98.70 | 99.96 |
| 4 | 101.11 | 100.64 |
| 5 | 100.81 | 101.97 |
| 6 | 101.57 | 100.64 |
| AVG | 100.71 | 100.2 |
| STDEV | 1.0688 | 1.2609 |
| %RSD | 1.06 | 1.26 |
LOD
Limit of detection was calculated by into Cobicistat and Darunavir method and LOD for Cobicistat was found to be 0.107 and Darunavir was0.333 respectively
LOQ
Limit of Quantification was calculated by into Cobicistat and Darunavir method and LOQ for Cobicistat and Darunavir were found to be 0.326 and 1.009 respectively
Robustness
Small deliberate changes in method like Flow rate, mobile phase ratio, and temperature are made but there were no recognized change in the result and are within range as per ICH Guide lines
Assay
Standard preparations are made from the API and Sample Preparations are from Formulation. Both sample and standards are injected six homogeneous samples. Drug in the formulation was estimated by taking the standard as the reference. The Average %Assay was calculated and found to be 100.71% and 100.2% for Cobicistat and Darunavir respectively
Conclusion
A simple, accurate, precise method was developed for the simultaneous estimation of the Cobicistat and Darunavir in tablet dosage form. Retention time of Cobicistat and Darunavir were found to be 3.170 min and 3.984 min. %RSD of the Cobicistat and Darunavir were and found to be 1.06 and 1.3 respectively. %Recover was Obtained as 100.71% and 100.2% for Cobicistat and Darunavir. LOD, LOQ values were obtained from regression equations of Cobicistat and Darunavir were 0.107 ppm, 0.326 ppm and 0.333 ppm, 1.009 ppm respectively. Regression equation of Cobicistat is y=10306x+1346, and of Darunavir is y=8883. x+2152. Retention times are decreased and that run time was decreased so the method developed was simple and economical that can be adopted in regular Quality control test in Industries.
References
- P. D. Sethi, Quantitative Analysis of Drugs in Pharmaceutical Formulations, CBS Publishers & Distributors, 1997.
- H. H. Willard, L. L. Merrit, J. A. Dean, F. A. Settle, Instrumental Method of Analysis, CBS Publishers & Distributors, 1986.
- R. A. Day, A. L. Underwood, Quantitative Analysis, PHI learning private limited, 2009.
- G. RamanaRao, S. S. N. Murthy, P. Khadgapathim, High performance liquid chromatography and its role in pharmaceutical analysis (Review), Eastern Pharm, 29 (1986), 53.
- R. S. Satoskar, S. D. Bhandarkar, S. S. Ainapure, Pharmacology and Pharmacotherapeutics, Popular Prakashan, 2001.
- Quantitative Analysis of Drugs in Pharmaceutical Formulations, CBS Publishers & Distributors
- Goodman, Gilman’s, The Pharmacological Basis of Therapeutics, McGraw-Hill health professions division, New york, 1996.
- F. Urooj, T. Mamatha, G. Rajesh, Novel RP-HPLC method development and validation of Cobicistat in bulk drug and tablet dosage form, Der Pharmacia Sinica, 5 (2014), 99-105.
- P. Rao, D. Reddy, D. Ramachandran, Stability indicating HPLC method for simultaneous estimation of emtricitabine, tenofovir, cobicistat and elvitegravir in pharmaceutical dosage form, World J Pharm Sci, 2 (2014), 1822-1829.
- S. Ganji, D. Satyavati, Development and validation of RP-HPLC method for the analysis of Cobicistat and related impurities in bulk and pharmaceutical dosage forms, Asian J Pharm Ana, 5 (2015), 1-8.
- K. Kalyani, V. Anuradha, Stability-indicating high performance liquid chromatographic method for the determination of Cobicistat, Int J Pharm Drug Anal, 4 (2012), 117-125.
- S. H. Rizwa, V. GirijaSastry, S. Gazi, Q. Imad, K. Bhameshan, A new and validated stability indicating rp-hplc analysis of darunavir and cobicistat in bulk drug and tablet dosage form, Int J Pharm Sci Rev Res, 36 (2016), 180-185.
- Bhavini Patel, Rp-hplc method development and validation for estimation of Darunavir ethanolate in tablet dosage, Int J Pharm Sci, 4 (2012), 1-8.
- B. Manisha, A. Mane , RP-HPLC method for determination of darunavir in bulk and pharmaceutical preparations, Asian J Pharm Ana, 5 (2018), 1-5.
- J. Chaves, R. Correa, Stability study of darunavir ethanolate tablets applying a new stability-indicating hplc method, 2013 (2013), 834173- 7.
- G. Raveendrababu, Development and validation of novel hplc method for estimation of darunavir in pharmaceutical formulations, Int J Res In Pharm Chem, 5 (2016), 1-9.
Copyright: © 2022 B Rajeswari, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

