Research Article: Journal of Drug and Alcohol Research (2023) Volume 12, Issue 4
Pharmacodynamics and Pharmacokinetics Drug Interactions between α-Mangostin and Gliclazide: A Pre-Clinical Study
Srinivasu Matta1 and Eswar Kumar Kilari2*2Department of Pharmacology, AU College of Pharmaceutical Sciences, India
Eswar Kumar Kilari, Department of Pharmacology, AU College of Pharmaceutical Sciences, India, Email: ekilari@gmail.com
Received: 03-May-2023, Manuscript No. JDAR-23-100826 ; Editor assigned: 05-May-2023, Pre QC No. JDAR-23-100826 (PQ); Reviewed: 19-May-2023, QC No. JDAR-23-100826; Revised: 24-May-2023, Manuscript No. JDAR-23-100826 (R); Published: 31-May-2023, DOI: 10.4303/JDAR/236239
Abstract
A study was conducted to investigate the impact of α-Mangostin on the pharmacodynamics and pharmacokinetics of gliclazide, a widely used drug for type II diabetes. The study was performed on normal rats, diabetic rats, and normal rabbits, with the administration of selected doses of gliclazide, α-Mangostin, and their combination. Appropriate washout periods were included between treatments. Blood samples were collected from rats and rabbits at regular intervals through retro-orbital and marginal ear vein punctures, respectively. All blood samples were analyzed for glucose using GOD/POD method, for serum insulin using ELISA method, and for gliclazide using HPLC. The results showed that gliclazide produced significant hypoglycemic and antihyperglycemic responses in normal/ diabetic rats and rabbits. The administration of a selected dose of α-Mangostin alone reduced blood glucose levels and improved the hypoglycemic response produced by gliclazide in combination, both in normal and diabetic rats. Similar effects were observed in normal rabbits, along with an enhancement of serum insulin levels. The pharmacokinetic parameters of gliclazide, such as AUC, AUMC, t1/2, Ke, Vdss clearance, and MRT, were significantly altered in normal rabbits when given in combination with α-Mangostin. This indicated changes in the metabolism and distribution pattern of gliclazide in the presence of α-Mangostin, potentially due to plasma protein displacement and inhibition of the CYP450 2C9 enzyme by α-Mangostin, leading to an increase in the plasma concentration of gliclazide. The study suggests that both single and multiple dose treatments may enhance the activity of gliclazide, and therefore, the therapy with the combination requires adjustment of dosing intervals between treatments.
Keywords
α-Mangostin; Gliclazide; Herbal drug interactions
Introduction
Herbal drug interactions are a crucial consideration for healthcare providers and patients alike. While many people utilize herbal supplements for various health benefits, they may not be aware of the potential interactions between these supplements and prescription or over-the-counter medications they may also be taking. These interactions can have severe consequences, including reduced medication efficacy, increased risk of side effects, and even toxicity [1]. It is therefore essential for healthcare providers to be knowledgeable about the possible interactions between herbal supplements and medications, and to educate their patients about the associated risks. Patients should also inform their healthcare provider of any herbal supplements they are using and should never mix supplements and medications without first consulting their healthcare provider. As herbal supplements gain popularity, it is important to research how they interact with other drugs. This research can inform healthcare providers in their treatment decisions, leading to better patient outcomes and safer healthcare [2]. Herbal medicines have several advantages, including antioxidant, cytoprotective, antidiabetic, anti-aging, and anti-inflammatory properties, making them a helpful addition to allopathic medicines for diabetics [3].
Diabetes mellitus is a condition where there is a long-term increase in blood glucose levels due to problems with insulin production or insulin resistance. It affects people worldwide and can lead to various complications. Antioxidants can help reduce the severity of these complications by combating oxidative stress [4,5]. Diabetes is becoming a significant problem worldwide, with an estimated 9.3% (463 million) of people affected in 2019. This number is expected to increase to 10.2% (578 million) by 2030 and 10.9% (700 million) by 2045 [6]. In India, the prevalence of diabetes has risen significantly in both rural and urban areas, from 2.4% and 3.3% in 1972 to 15.0% and 19.0%, respectively, between 2015 and 2019 [7]. Herbal supplements are often used as a complementary or alternative treatment for diabetes and other health conditions. However, they can interact with diabetes medications and affect blood sugar levels, leading to adverse effects such as low or high blood sugar. This is important to consider as many people with diabetes use herbal supplements, and the condition affects millions worldwide [8]. Many herbal supplements used for diabetes are not well-studied for safety or effectiveness, making it hard for healthcare providers to predict potential interactions with diabetes medication [9]. In summary, it’s important for diabetics to manage their herbal drug interactions properly to avoid negative effects. Healthcare providers need to know about potential interactions, inform patients about the risks, and advise patients to talk to them before using any herbal supplements.
Α-mangostin is a natural compound found in the pericarps, bark, and sap of the mangosteen tree, known for its diverse range of beneficial properties. Research has shown that Α-mangostin exhibits antioxidant, anti-obesity, anti- parasitic, anti-allergic, antiviral, antibacterial, anti-inflammatory, and anticancer effects [10]. It is commercially available in various herbal products such as capsules, juice concentrates, and supplements, including brands like Xango Reserve and Dynamic Health Mangosteen Gold. These products are recognized for their potent antioxidant and anti-inflammatory properties, as well as their potential to aid in diabetes management. Notably, α-Mangostin has been found to stimulate insulin production and safeguard pancreatic beta cells from oxidative damage caused by free radicals. Moreover, studies in diabetic rats have demonstrated a dose-dependent reduction in blood glucose levels, along with alterations in the activity of key enzymes involved in glucose metabolism [11]. With the global mangosteen market projected to experience substantial growth, as reported by DataM Intelligence in 2022 it becomes crucial to understand the anti-diabetic and hypoglycemic effects of α-Mangostin, as they may potentially influence the pharmacodynamics of the anti-diabetic medication, Gliclazide [12].
Established activities such as plasma protein displacement, P-glycoprotein (Pgp) inhibition, and CYP enzyme inhibition can potentially impact the pharmacokinetic properties of gliclazide. An in vitro study has demonstrated that α-Mangostin inhibits multiple cytochrome P450 isoforms, including CYP2C9, which plays a crucial role in the metabolism of gliclazide [13]. Furthermore, α-Mangostin exhibits additional properties, including the potential to inhibit Pgp, modify Pgp expression, and display α-glucosidase inhibitory activity [14,15]. Studies have indicated that α-Mangostin has a high protein binding capacity, with binding values ranging from 58.5% to 96.66% in plasma [16]. Given that gliclazide is also a plasma protein-bound drug, there is a possibility of interaction between the two. The combination of herbal products containing α-Mangostin and the commonly used drug gliclazide may result in a drug-herbal interaction. Therefore, this study aimed to investigate this interaction.
Materials and Methods
Drugs and chemicals
Gliclazide was acquired as a gift sample from Wockhardt Pharmaceuticals, Aurangabad, while α-Mangostin was gifted by Laila Impex, Vijayawada, Andhra Pradesh. Streptozotocin was procured from Sigma Aldrich. All reagents and chemicals utilized in the study were of analytical grade.
Animals: The study utilized inbred adult male Albino Wistar rats, aged 8 weeks to 9 weeks and weighing between 170 g to 250 g, as well as Albino rabbits that were 3 months old and weighed between 1 kg to 1.5 kg, of either sex. These animals were obtained from Mahaveer Enterprises, Hyderabad, India and were maintained under standard laboratory conditions with an ambient temperature of 25°C ± 2°C and 50% ± 15% relative humidity, with a 12-hour light/12-hour dark cycle. They were provided with a commercial pellet diet (Rayan’s Biotechnologies Pvt. Ltd., Hyderabad, India) and water ad libitum. The experimental protocol was approved by the Institutional Animal Ethics Committee and the regulatory body of the government, with registration number 516/01/A/CPCSEA. Prior to the experiment, the animals were fasted for 18 hours, with access to water, and during the experiment, both food and water were withdrawn.
Experimental design
In this study, it was found that a dose of 50 mg/kg of α-Mangostin resulted in an optimal oral hypoglycemic effect, and was therefore chosen as the study dose for both rats and rabbits. To investigate the relationship between gliclazide dose and effect, normal rats and rabbits were used. Based on the results, doses of 1 mg/kg and 5.6 mg/kg body weight were selected for administration to rats and rabbits, respectively. The α-Mangostin was administered orally in a formulation prepared by suspending it with 0.5% carboxymethyl cellulose Na. On the other hand, the gliclazide was administered as a solution that was made by dissolving it in a small amount of 0.1 N NaOH and then adding water to achieve the desired volume. The study design is described below.
Stage 1: Pharmacodynamic interaction in normal rats [17]
Stage 2: Pharmacodynamic interaction in diabetic rats [17]
Stage 3: Pharmacodynamic and pharmacokinetic interaction in normal rabbits [18]
Stage 1-pharmacodynamic interaction in normal rats: In the stage 1 experiment, 6 rats were utilized and subjected to periodic treatments with a 1-week washout period. The rats were initially administered with 1 mg/kg gliclazide orally, followed by blood sample collection at predetermined time intervals. Subsequently, a 1 week washout period was given. Similar treatments were administered with either orally administered α-Mangostin or a combination treatment with both α-Mangostin and gliclazide at the aforementioned doses. Blood samples were collected at predetermined time intervals subsequent to each treatment.
Stage 2-pharmacodynamic interaction in diabetic: In the stage 2 experiment, diabetes was induced in rats through the intraperitoneal injection of a single dose of streptozotocin (65 mg/kg) [19]. Rats exhibiting blood glucose levels exceeding 250 mg/dl were selected for inclusion in the study. The identical treatment protocols outlined in stage 1 were administered to the diabetic rats.
Stage 3-pharmacodynamic and pharmacokinetic interaction in normal rabbits: In the stage 3 experiment, 6 rabbits were utilized. The rabbits were administered gliclazide via the oral route at a dose of 5.6 mg/kg body weight, and blood samples were collected at predetermined time points. Following a 1 week washout period between experiments, the same procedure was conducted with either orally administered α-Mangostin or a combination treatment with both α-Mangostin and gliclazide at the same previously stated doses. After the single-dose interaction study, the same rabbits received daily treatments with α-Mangostin for 7 consecutive days with regular feeding. On day 8, the rabbits fasted for 12 hours before being given α-Mangostin. After 30 minutes, the rabbits were administered gliclazide at a dose of 5.6 mg/kg body weight. Blood samples were collected at predetermined intervals following each treatment of gliclazide, α-Mangostin, or combination treatments for both single and multiple treatments.
Blood sampling
Blood samples were collected from the retro-orbital plexus of rats at 0 hours, 1 hours, 2 hours, 4 hours, 6 hours, 8 hours, 12 hours, 18 hours, and 24 hours. For rabbits, blood samples were withdrawn from the marginal ear vein at 0 hours, 1 hours, 2 hours, 4 hours, 6 hours, 8 hours, 12 hours, 16 hours, 20 hours and 24 hours. The rat and rabbit blood samples were tested for blood glucose using the GOD/POD method in a semi-auto analyser [20]. Additionally, the rabbit blood samples were analyzed for serum gliclazide using the HPLC method [18]. For the estimation of serum insulin, rabbit blood samples at 0 hours, 1 hours, 4 hours, 8 hours, 16 hours, and 24 hours were used, and the ELISA method was employed [21].
Data and Statistical Analysis
Pharmacokinetic parameters were calculated using Ramkin software. Data are expressed as mean ± standard error of mean values. Student’s paired t-test was used as statistical tool and p<0.05 is considered as statistically significant.
Results
Gliclazide demonstrated hypoglycemic activity in normal rats, with a maximum biphasic reduction of 34.87% ± 0.92% and 36.70% ± 0.82% observed at 2-hour and 8-hour intervals, respectively (Table 1). In diabetic rats, gliclazide exhibited antihyperglycemic activity, with a peak biphasic reduction of 44.48% ± 0.41% and 44.56% ± 0.69% at 2-hour and 12-hour intervals, respectively (Table 2). Additionally, in rabbits, the peak hypoglycemic activity was observed with a maximum reduction of 39.27% ± 0.50% at the 4-hour time interval (Tables 3-5).
Table 1: Mean percent blood glucose reduction of gliclazide in presence and absence of a Mangostin in normal rats (n=6)
| Time (h) | a Mangostin | Gliclazide | Gliclazide+a Mangostin |
|---|---|---|---|
| 1 | 18.16 ± 0.99 | 25.09 ± 1.38 | 26.21 ± 1.35ns |
| 2 | 25.13 ± 0.64 | 34.87 ± 0.92 | 37.85 ± 2.56ns |
| 4 | 31.45 ± 0.67 | 32.27 ± 0.58 | 47.73 ± 1.42* |
| 6 | 26.32 ± 1.01 | 30.77 ± 0.69 | 43.28 ± 0.57* |
| 8 | 20.42 ± 0.76 | 36.70 ± 0.82 | 39.96 ± 0.95* |
| 12 | 15.83 ± 0.62 | 26.67 ± 1.48 | 35.36 ± 0.62* |
| 18 | 12.06 ± 0.69 | 19.47 ± 1.58 | 31.73 ± 0.45* |
| 24 | 8.99 ± 0.50 | 11.31 ± 1.14 | 21.65 ± 1.25* |
| Note: Data expressed as mean ± standard deviation *Statistically significant when compared with gliclazide control |
|||
Table 2: Mean percent blood glucose reduction of gliclazide in presence and absence of a-Mangostin in diabetic rats (n=6)
| Time (h) | a-Mangostin | Gliclazide | Gliclazide+a-Mangostin |
|---|---|---|---|
| 1 | 27.24 ± 1.07 | 34.70 ± 1.03 | 43.80 ± 0.79* |
| 2 | 30.78 ± 0.98 | 44.48 ± 0.41 | 54.26 ± 0.93* |
| 4 | 34.94 ± 1.26 | 40.56 ± 0.84 | 55.45 ± 0.98* |
| 6 | 36.57 ± 1.48 | 36.87 ± 0.98 | 56.16 ± 0.80* |
| 8 | 32.23 ± 1.12 | 40.36 ± 0.35 | 54.17 ± 0.65* |
| 12 | 28.63 ±1.11 | 44.56 ± 0.69 | 49.09 ± 0.93* |
| 18 | 25.18 ±1.25 | 38.86 ± 0.78 | 45.88 ± 0.70* |
| 24 | 22.37 ±1.27 | 32.14 ± 0.85 | 42.13 ± 0.96* |
| Note: Data expressed as mean ± standard deviation *Statistically significant when compared with gliclazide control |
|||
Table 3: Mean percent blood glucose reduction of gliclazide in presence and absence of a-Mangostin in rabbits (n=6)
| Time (h) | a-Mangostin | Gliclazide | Gliclazide+a-Mangostin SDT | Gliclazide + a-Mangostin MDT |
|---|---|---|---|---|
| 1 | 20.66 ± 0.47 | 26.26 ± 0.92 | 35.52 ± 1.07* | 37.33 ± 0.97* |
| 2 | 27.01 ± 0.90 | 35.06 ± 0.71 | 47.29 ± 1.15* | 46.25 ± 1.50* |
| 4 | 33.34 ± 1.11 | 39.27 ± 0.50 | 51.55 ± 1.62* | 49.95 ± 0.81* |
| 6 | 28.70 ± 1.10 | 35.23 ± 0.29 | 51.41 ± 0.82* | 52.10 ± 0.91* |
| 8 | 22.55 ± 1.40 | 29.99 ± 0.78 | 49.14 ± 1.33* | 53.28 ± 0.86* |
| 12 | 17.51 ± 1.36 | 25.95 ± 1.18 | 44.86 ± 1.46* | 49.94 ± 1.10* |
| 16 | 13.66 ± 1.33 | 20.88 ± 1.29 | 40.44 ± 1.34* | 45.94 ± 1.24* |
| 20 | 9.17 ± 1.35 | 15.48 ± 1.69 | 37.00 ± 1.32* | 38.13 ± 2.02* |
| 24 | 5.32 ± 0.93 | 9.42 ± 1.47 | 31.43 ± 1.56* | 33.80 ± 2.16* |
| Note: Data expressed as mean ± standard deviation *Statistically significant when compared with gliclazide control |
||||
Table 4: Mean change in serum insulin levels (µIU/mL) of gliclazide in presence and absence of a Mangostin in rabbits (n=6)
| Time (h) | a-Mangostin | Gliclazide | Gliclazide+a Mangostin SDT | Gliclazide+a Mangostin MDT |
|---|---|---|---|---|
| 0 | 9.35 ± 0.16 | 9.25 ± 0.28 | 9.58 ± 0.16ns | 9.63 ± 0.21ns |
| 1 | 10.58 ± 0.19 | 10.95 ± 0.26 | 12.09 ± 0.08ns | 11.98 ± 0.12ns |
| 4 | 11.87 ± 0.13 | 12.84 ± 0.12 | 14.61 ± 0.24* | 15.22 ± 0.19* |
| 8 | 10.75 ± 0.09 | 11.40 ± 0.13 | 14.12 ± 0.22* | 15.91 ± 0.16* |
| 16 | 10.30 ± 0.16 | 10.52 ± 0.11 | 12.25 ± 0.13* | 14.93 ± 0.27* |
| 24 | 9.44 ± 0.19 | 9.92 ± 0.15 | 11.04 ± 0.12* | 13.54 ± 0.28* |
| Note: Data expressed as mean ± standard deviation. *Statistically significant when compared with gliclazide control Abbreviations-MDT: Multiple-Dose Treatment; SDT: Single-Dose Treatment |
||||
Table 5: Mean pharmacokinetic parameters of gliclazide before and after administration of a-Mangostin in rabbits (n=6)
| Time (h) | Gliclazide | Gliclazide+a-Mangostin SDT | Gliclazide+a-Mangostin MDT |
|---|---|---|---|
| AUC0-24 (µg/ml/hr) | 4823.70 ± 295.394 | 7822.26 ± 584.765* | 9298.74 ± 387.123* |
| AUC0-24 (µg/ml/hr*h) | 52329.70 ± 11294.470 | 126280.50 ± 13540.310* | 158081.90 ± 9732.209* |
| Kel (h-1) | 6.69 ± 0.431 | 4.39 ± 0.462ns | 3.46 ± 0.782ns |
| AUC0-a (µg/ml/hr) | 6265.76 ± 524.211 | 12450.54 ± 1292.791* | 16034.67 ± 1241.383* |
| AUMC0-a (µg/ml/hr*h) | 118598.90 ± 18365.780 | 355916.00 ± 59312.390* | 511071.20 ± 81444.250* |
| t½ (h) | 10.60 ± 0.715 | 16.58 ± 1.535ns | 17.94 ± 2.065ns |
| Ka (h-1) | 1.15 ± 0.000 | 1.15 ± 0.000 | 1.15 ± 0.000 |
| Clearance (ml/hr) | 1393.32 ± 116.292 | 706.57 ± 85.935* | 545.35 ± 51.822* |
| Clearance (ml/hr/kg) | 924.42 ± 74.077 | 476.47 ± 52.591* | 359.49 ± 31.337* |
| Vd SS (ml) | 23871.22 ± 1211.749 | 18072.13 ± 947.333ns | 15826.60 ± 761.815* |
| Vd SS (ml/kg) | 15875.70 ± 848.239 | 12275.43 ± 800.487ns | 10447.48 ± 431.076* |
| Vd area (ml) | 20811.70 ± 1036.709 | 16092.35 ± 892.852ns | 13747.75 ± 741.969* |
| Vd area (ml/kg) | 13839.07 ± 723.945 | 10952.95 ± 824.508ns | 9076.59 ± 451.349* |
| MRT (h) | 18.46 ± 1.371 | 27.66 ± 2.327ns | 30.93 ± 2.762* |
| C max (µg/ml) | 384.95 ± 16.400 | 497.25 ± 27.550* | 615.76 ± 26.320* |
| T max (h) | 4.00 ± 0 | 4.00 ± 0.000ns | 4.00 ± 0.000ns |
| Note: Data expressed as mean ± standard deviation. Abbreviations: AUC: Area Under the Concentration Time Curve; AUMC: Area Under First Moment Curve; CL: Clearance; Cmax: Peak serum concentration; Kel: Elimination Rate Constant; MDT: Multiple-Dose Treatment; MRT: Mean Residence Time; SD: Standard Deviation; SDT: Single-Dose Treatment; Tmax: Peak time; T1/2: Terminal Half-Life |
|||
α-Mangostin alone significantly reduced the blood glucose levels of normal rats, diabetic rats, and rabbits between 4 hours and 6 hours. In the single-dose combination study, a significant decrease in blood glucose levels was observed in normal rats (47.73% ± 1.42%) and diabetic rats (56.16% ± 0.80%). Moreover, in rabbits, a peak decrease in blood glucose (51.55% ± 1.62%) was accompanied by a simultaneous increase in both insulin (14.61 μIU/mL ± 0.24 μIU/ mL) and serum gliclazide levels (497.25 ng/ml ± 27.55 ng/ ml) at the 4th hour. In the multiple-dose study, a similar pattern was observed at the 8th hour, with corresponding increases in blood glucose (53.28% ± 0.86%), serum insulin (15.91 μIU/mL ± 0.16 μIU/mL), and serum gliclazide levels (457.10 ng/ml ± 21.90 ng/ml) at 4th hour.
Discussion
Drug interactions are commonly observed in clinical practice and animal experiments, and the mechanisms of these interactions are often assessed using animal models [22,23]. In this study, The impact of α-Mangostin on the pharmacodynamics of gliclazide was examined in both normal and diabetic rats, as well as in normal rabbits. Additionally, the pharmacokinetics of gliclazide was specifically investigated in normal rabbits. The use of the normal rat model helped validate the observed response of the drug, which is relevant to its actual usage in type II diabetes [18]. Rabbit model represents a different species and validates the results.
Commercially, α-Mangostin is marketed in various forms as an immune enhancer and nutritional supplement. It offers numerous health benefits, particularly for diabetics. It may reduce blood glucose levels by enhancing insulin sensitivity in both normal and diabetic rats [11]. Single-dose administrations of α-Mangostin alone and in combination with gliclazide alter blood glucose levels in both normal and diabetic rats, indicating the presence of an interaction. The findings in diabetic and normal rats demonstrate that there is a chance of interaction among rats (a rodentspecies). Moreover, normal rabbits treated with gliclazide experienced changes in their hypoglycemic effects due to α-Mangostin, proving that non-rodent animals can also be affected by this drug-drug interaction. Significant changes were also observed in pharmacokinetic parameters such as AUC, AUMC, Clearance, Vdss, Vdarea, and Cmax of gliclazide with both single and multiple dose treatments of α-Mangostin. The increase in AUMC suggests an improved availability of gliclazide in the presence of α-Mangostin. However, this increased bioavailability cannot be attributed to improved absorption since the absorption rate and absorption half-life of gliclazide remained unaltered (Figure 1).
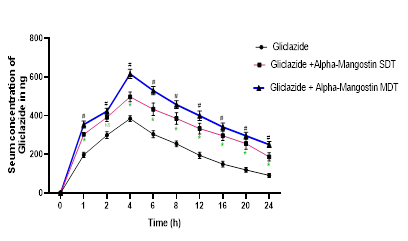
Figure 1: Mean gliclazide concentration in serum (ng/mL) before and after treatment with a Mangostin in rabbits (n=6)
Gliclazide is a drug that exhibits high protein binding (85%- 97%) [24]. α-Mangostin also demonstrates a mean plasma protein binding of 91.8% ± 13.5% in humans [16,25]. Consequently, there is a greater possibility of α-Mangostin displacing gliclazide from its protein-bound sites. In the experiment results, the increase in gliclazide blood levels in the presence of α-Mangostin is due to plasma protein displacement or metabolism inhibition but not due to an increase in absorption, as Ka and Tmax (4 hours) have not been altered. Moreover, Gliclazide is primarily metabolized by hepatic CYP450 2C9 and CYP450 3A4. An in vitro study has proven that the herbal isolate α-Mangostin inhibits hepatic CYP450 2C9 [13]. Hence, there is a possibility of interaction at the hepatic metabolism of drugs, resulting in reduced gliclazide metabolism and raised serum levels in the presence of α-Mangostin, leading to an increased gliclazide effect. Therefore, the study results showing the rise in blood levels of gliclazide in the presence of α-Mangostin may be because of the combined influence of competitive inhibition of metabolism and plasma protein displacement.
Conclusion
The enhancement in gliclazide activity in the presence of selected herbal isolates was statistically significant but not pharmacologically significant, as there was only a marginal increase in gliclazide response and no hypoglycemic convulsions were observed. The aforementioned phenomenon indicates the safety of their combinations. Since the combinations were found to be safe in 2 different species, they are likely to be safe clinically. In fact, no adverse cases have been reported clinically.
Acknowledgement
The authors thank Wockhardt pharmaceuticals, Aurangabad and RPG Laila Impex, Vijayawada for providing the gift sample of gliclazide and α-Mangostin respectively.
Conflict Of Interest
Authors have no conflict of interest to declare.
References
- S. Hussain, Patient counseling about herbal-drug interactions, African J Tradit Complement Altern Med, 8(2011):152-63.
- M. Ekor, The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety, Front Pharmacol, 4(2014):177-187.
- W. Kooti, M. Farokhipour, Z. Asadzadeh, D. Ashtary-Larky, M. Asadi-Samani, The role of medicinal plants in the treatment of diabetes: A systematic review, Electron physician, 8(2016): 1832-1842.
- D. Ziegler, P.A. Low, W.J. Litchy, A.J.M. Boulton, A.I. Vinik, et al. Efficacy and safety of antioxidant treatment with a-lipoic acid over 4 years in diabetic polyneuropathy, Diabetes Care, 34(2011):2054-2060.
- B. Lipinski, Pathophysiology of oxidative stress in diabetes mellitus, J Diabetes Complications, 15(2001):203-210.
- P. Saeedi, I. Petersohn, P. Salpea, B. Malanda, S. Karuranga, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition, Diabetes Res Clin Pract, (2019):157-07843.
- P. Ranasinghe, R. Jayawardena, N. Gamage, N. Sivanandam, A. Misra, Prevalence and trends of the diabetes epidemic in urban and rural India: A pooled systematic review and meta-analysis of 1.7 million adults, Ann Epidemiol, 58(2021):128-148.
- A. Pandey, P. Tripathi, R. Pandey, R. Srivatava, S. Goswami, Alternative therapies useful in the management of diabetes: A systematic review, J Pharm Bioallied Sci, 3(2011):504-512.
- L.V. Cross, J.R. Thomas, Safety and efficacy of dietary supplements for diabetes, Diabetes Spectr, 34(2021):67-72.
- M.Y. Ibrahim, N.M. Hashim, A.A. Mariod, S. Mohan, M.A. Abdulla, et al. a-Mangostin from Garcinia mangostana Linn: An updated review of its pharmacological properties, Neuropsychopharmacology, 9(2016):317-329.
- D. Lee, Y.M. Kim, K. Jung, Y.W. Chin, K. Kang, Alpha-Mangostin improves insulin secretion and protects ins-1 cells from streptozotocin-induced damage, Int J Mol Sci, 19(2018):1484-1497.
- DataM Intelligence, Mangosteen market size, competitive landscape and market forecast-2029, 2022.
- R.S. Foti, J.T. Pearson, D.A. Rock, J.L. Wahlstrom, L.C. Wienkers, In vitro inhibition of multiple cytochrome p450 isoforms by xanthone derivatives from mangosteen extract, Drug Metab Dispos, 37(2009):1848-55.
- P. Dechwongya, S. Limpisood, N. Boonnak, S. Mangmool, M. Takeda-Morishita, et al. The intestinal efflux transporter inhibition activity of xanthones from mangosteen pericarp: An in silico, in vitro and ex vivo approach, Molecules, 25(2020):5877-5891.
- F.M. Djeujo, V. Francesconi, M. Gonella, E. Ragazzi, M. Tonelli, et al. Anti-a-gucosidase and antiglycation activities of a-mangostin and new xanthenone derivatives: Enzymatic kinetics and mechanistic insights through in vitro studies, Molecules 27(2022):547.
- S.Y. Han, B.H. You, Y.C. Kim, Y.W. Chin, Y.H. Choi, Dose-independent ADME properties and tentative identification of metabolites of a-mangostin from garcinia mangostana in mice by automated microsampling and UPLC-MS/MS methods, PLoS One, 10(2015):e0131587-e0131603.
- S. Satyanarayana, E.K. Kilari, Influence of nicorandil on the pharmacodynamics and pharmacokinetics of gliclazide in rats and rabbits, Mol Cell Biochem, 291(2006):101-105.
- K. Eswar Kumar, S. Mastan, Influence of efavirenz and nevirapine on the pharmacodynamics and pharmacokinetics of gliclazide in rabbits, J Endocrinol Metab, 1(2011):113-124.
- B.L. Furman, Streptozotocin-induced diabetic models in mice and rats, Curr Protoc Pharmacol, 70(2015):1-20.
- V.N. Ambade, Y. Sharma, B. Somani, Methods for estimation of blood glucose : A comparative evaluation, Med J Armed Forces India, 54(1998):131-133.
- R.M. Lequin, Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA), Clin Chem, 51(2005):2415-2418.
- C. Palleria, A. Di Paolo, C. Giofrè, C. Caglioti, G. Leuzzi, et al. Pharmacokinetic drug-drug interaction and their implication in clinical management, J Res Med Sci, 18(2013):601-10.
- R.C. Gupta, Drug metabolism studies in animal models, Indian J Pharmacol, 32(2000):62-66.
- N. Seedher, M. Kanojia, Reversible binding of antidiabetic drugs, repaglinide and gliclazide, with human serum albumin, Chem Biol Drug Des, 72(2008):290-296.
- S. Hidayat, F.M. Ibrahim, K.F. Pratama, M. Muchtaridi, The interaction of a-mangostin and its derivatives against main protease enzyme in COVID-19 using in silico methods, J Adv Pharm Technol Res, 12(2021):285-290.
Copyright: © 2023 Eswar Kumar Kilari, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

