Research Article: Journal of Drug and Alcohol Research (2022) Volume 11, Issue 6
Lobaria pulmonaria (L.) Hoffm. Attenuates Alcoholic Liver Injury in Wistar Rats by Reducing Oxidative Stress
Kishore Naidu Killari1*, Ho Viet Hieu2, Prasad Konduri1, Manasa Rampathoti3, Santosh Kumar Ranajith4 and Mallikarjuna Rao Talluri52Department of Medical Microbiology and Parasitology, Duy Tan University, Vietnam
3Department of Pharmacology, JITS College of Pharmacy, India
4Department of Pharmacology, Centurion University of Technology and Management, India
5ACR Laboratories, India
Kishore Naidu Killari, Department of Pharmacology, Shri Vishnu College of Pharmacy, India, Email: kishorenaidu.killari@gmail.com
Received: 27-May-2022, Manuscript No. jdar-22-65174; Editor assigned: 30-May-2022, Pre QC No. jdar-22-65174 (PQ); Reviewed: 13-Jun-2022, QC No. jdar-22-65174; Revised: 20-Jun-2022, Manuscript No. jdar-22-65174 (R); Published: 27-Jun-2022, DOI: 10.4303/jdar/236181
Abstract
Background: Lichen, Lobaria pulmonaria (L.) Hoffm. belong to the family Lobariaceae. L. pulmonaria is being traditionally used to cure lung ailments, liver diseases, diarrhoea, heavy menstrual flow, and jaundice. With such a wide range of medicinal applications, it’s essential to scientifically authenticate traditional usage of L. pulmonaria. The present study is aimed to evaluate the antioxidant and hepatoprotective properties of selected ethanolic fractions of L. pulmonaria in alcohol induced oxidative stress in rats.
Methods: In vitro antioxidant activities were performed using DPPH radical scavenging and ferric ions reducing power assays. In vivo hepatoprotective activity was assessed by using ethanol induced oxidative stress in Wistar rats.
Results: Initially, ethanolic extract of L. pulmonaria was fractionated using column chromatography. The preliminary antioxidant screening of these fractions identified two main bioactive fractions (LP3 and LP4), which were found to have significant radical scavenging and metal ion chelation properties compared with ascorbic acid. Based on the antioxidant profile, LP3 and LP4 were evaluated for hepatoprotective activity in ethanol intoxicated rats. The Wistar rats were grouped (n=6) and treated with LP3 and LP4 (100 and 200 mg/kg), ethanol (5 g/kg, 20%w/v) and silymarin (100 mg/kg) orally for 28 days. The outcomes of the study found that chronic administration of ethanol significantly (P<0.0001) altered the liver parameters and oxidative stress markers (MDA, SOD, and CAT). The co-administration of LP4 prominently ameliorated the oxidative stress induced by ethanol compared to LP3. Histopathological studies further supported the significant protective action of LP4.
Conclusion: The present study demonstrates that the L. pulmonaria possess significant antioxidant properties by augmenting the magnitude of the antioxidant enzymes SOD and CAT and further reducing MDA levels.
Keywords
Antioxidant; Ethanol-induced; Hepatoprotective; Oxidative stress; Lobaria pulmonaria.
Introduction
Liver is a major organ in our body that regulates various physiological functions and almost about 500 essential tasks are attributed to liver. Hence, the damage or loss of any liver function can have serious effects on our health. According to recent statistics, liver illnesses kill over two million people each year throughout the world. Cirrhosis complications account for almost one million deaths. The remaining are mainly by viral hepatitis and hepatocellular carcinoma [1,2].
The alteration in the liver’s normal functioning led to abnormal metabolism and decreased the elimination of toxic metabolites that alters the homeostasis. The main factors causing liver damage are disorders/diseases, drugs, chronic alcoholism, and various poisonous substances [1]. Repeated liver injury by toxicants, synthetic drugs, or hepatic viruses can cause sustained inflammation and wound healing processes that result in the accumulation of excess extracellular matrix that does not undergo degradation leading to the fibrosis of the liver tissue. The accumulated excess extracellular matrix proteins damage the liver by forming a fibrous scar leading to liver cirrhosis and cancer. The management of inflammation, oxidative damage, apoptosis, and related tissue responses are the essential therapeutic strategies for preventing liver failure [3,4].
Day by day, alcohol consumption is increasing in all countries across the world. According to the World Health Organization, 3.3 million deaths related to chronic alcoholism have been recorded every year globally [5]. Generally, 80% of consumed alcohol is metabolized by cytochrome P450 enzyme resulting in the generation of the toxic metabolite named acetaldehyde. The accumulated acetaldehyde leads to the production of reactive oxygen free radicals (ROS), which ultimately culminates in oxidative stress and tissue inflammation [5,6].
The exact mechanism of alcohol-induced hepatotoxicity is complicated and unclear. But, it is proposed by few researchers that the metabolites of alcohol alter the NAD/ NADPH ratio, increased generation of ROS, damaging the DNA, proteins, and mitochondrial respiratory chain, oxidation of lipids, activation of kuffer cells and metabolism of iron [7-9]. Hence, there is a need to search for multi-targeted therapy against alcohol induced intoxication.
Lichen, Lobaria pulmonaria (L.) Hoffm (family: Lobariaceae) is widely distributed in Asia and Europe. Traditionally, lichen L. pulmonaria used to treat lung ailments, liver diseases, diarrhoea, heavy menstrual flow, and jaundice [10]. Taken together, the present study aimed to evaluate the antioxidant and hepatoprotective activities of bioactive fractions of ethanol extract of L. pulmonaria against chronic ethanol induced intoxication in Wistar albino rats.
Materials and Methods
Lichen material
Lichen L. pulmonaria were collected from Nilagiri hills, Tamil Nadu, India (6070 feet above sea level), in August 2019. The material was authenticated and deposited in the Bryophyte herbarium, CSIR-National Botanical Research Institute (NBRI), Lucknow, India, with a voucher specimen (No. 42743/2019).
Extraction and fractionation
The shade dried L. pulmonaria were ground into a coarse powder using an electrical blender. By hot continuous percolation technique, the powdered material (500 g) was extracted with ethanol for 48 h using a Soxhlet apparatus [11,12]. The obtained solvent mixture was concentrated under reduced pressure using rotavapor (Buchi R-210 Rotavapor, Marshall Scientific, USA) yielded ethanolic extract of L. pulmonaria (LP, 20 g, 4% w/w) as dark greenish solid. Using column chromatography of mesh size 100-200, LP (30 g) was fractionated using a hexane/ethyl acetate solvent system (step gradient flow from 100:0, 95:5, 90:10, …, 5:95, 0:100), which yielded five fractions (LP1-4) [13,14]. These fractions were subjected to antioxidant activity [15,16] (as per the procedure mentioned below) and noticed that only two of them, namely LP3 (10.5 g) and LP4 (9.8 g), were biological more active (Table 1).
Table 1: Primary screening of antioxidant activity of LP and its fractions (LP1-LP4).
| Sample | Percentage inhibition (%) at 100 µg/ml# | |
|---|---|---|
| DPPH radicals | Ferric ions | |
| LP | 79.08 ± 1.61 | 79.15 ± 1.92** |
| LP1 | 10.19 ± 0.54*** | 12.47 ± 0.67*** |
| LP2 | 12.15 ± 1.01*** | 10.02 ± 1.09*** |
| LP3 | 62.15 ± 2.20*** | 75.85 ± 4.18** |
| LP4 | 72.02 ± 3.09*** | 78.09 ± 3.44** |
| Ascorbic acid | 99.98±0.02 | 99.97 ± 0.03 |
| #mean ± SD (n=3); P**<0.001, P***<0.0001, as compared with standard group using one-way ANOVA with Student-Newman-Keuls post hoc test | ||
In vitro antioxidant activity
Evaluation of DPPH radical scavenging activity: By employing the 1,1-diphenyl-2-picrylhydrazyl (DPPH, Sigma Aldrich Co., USA) assay in triplicate, the LP1-LP4 and LP were evaluated for antioxidant activity [15]. To the known concentrations of LP1-LP4 and LP added 0.004% DPPH, and incubated for 30 min at 37°C. Later, absorbance was measured at 517 nm against the blank. Ascorbic acid was used as a standard drug.
Evaluation of ferric ion reducing power assay: The ferric ion reducing power assay was determined in triplicate by the modified method of Haritha et al. [16]. To 2.5 ml of potassium ferricyanide added various concentrations of LP1- LP4 and LP and incubated at 50°C for 20 min. To it, 0.5 ml of ferric chloride (0.1%) and 2.5 ml trichloroacetic acid (10%) were added, and the absorbance was noted at 700 nm. Ascorbic acid was used as a standard drug.
Total phenol and flavonoid contents
The total flavonoid and phenolic content of the extract were evaluated by using aluminum chloride and Folin-Ciocalteau reagent, respectively [17,18]. The total flavonoid and phenolic content of the LP3 and LP4 were expressed as rutin and gallic acid equivalent, respectively.
Animals
Adult Wistar albino rats (weighing 190 ± 10 g, age 6-8 weeks) of either sex were used in this study. The animals were given food and water ad libitum and were housed in the Animal House of the Vishnu College of Pharmacy under the standard condition with a temperature of 25 ± 2°C, the relative humidity (50 ± 10%), and a 12 h light/12 h dark cycle. This study was approved by the Institutional Animal Ethics Committee (Code: 12/IAEC/SVCP/2020-21).
Acute toxicity studies
The OECD main test 423 was utilized for acute toxicity studies. Rats were randomly divided into four groups (n=3) and dosed with 5, 50, 300 and 2000 mg/kg body weight (b.w) of LP suspended in gum acacia and tween-80. The test animals have undergone fasting overnight before administering the LP using oral gavage. The testing was ended until the last three animals survived the upper bound dose, and all of the test animals were observed up to 14 days [19,20].
Experimental protocol of hepatoprotective activity
At the beginning of the experiment, rats were randomly divided into seven groups (six rats in each group). In group 1 (normal control), rats were administered orally with only 0.2 ml of the vehicle for 28 days. In group 2 (toxic control), rats were dosed orally with ethanol (5 g/kg, 20%w/v) for 28 days. In group 3 (standard), rats were received 100 mg/ kg b.w of silymarin and ethanol (5 g/kg, 20%w/v) orally for 28 days. Rats in groups 4 and 5 received 100 mg/kg b.w (as a low dose) and 200 mg/kg b.w (as a high dose) of LP3 and ethanol (5 g/kg, 20%w/v), respectively, orally for 28 days. Rats in groups 6 and 7 received 100 mg/kg b.w (as a low dose) and 200 mg/kg b.w (as a high dose) of LP4 and ethanol (5 g/kg, 20%w/v), respectively, orally for 28 days.
Collection of serum samples
The blood samples were obtained from the portal vein 0.5 ml of blood samples were transferred into laboratory tubes containing pre-autoclaved nutrient broth medium (Sigma- Aldrich, Germany) and put in an incubator at 37°C. The remaining blood samples decanted gently into collection plastic tubes, centrifuged at 4000 rpm for 5 min. Then serum was obtained, aliquoted into microtubes, and stored at -80°C for biochemical analysis [21].
Tissue homogenization
At the end of the study, by cervical dislocation, all the rats were sacrificed. The livers were removed, weigh up, and washed thoroughly. Some portion of tissue was stored immediately in buffered formalin (10%) for histopathological studies, and the remaining tissue was processed. In 0.05 M of ice-cold phosphate buffer saline (pH 7), the tissue was minced into small pieces and homogenized with a Homogenizer (Remi Homogenizer, Mumbai, India) to obtain 10% whole homogenate. To the homogenate, an equal volume of trichloroacetic acid (10%) was mixed and centrifuged (Sigma-3-30 KS, USA) for 10 min at 5000 rpm [22].
Assessment of oxidative stress parameters
The above obtained supernatant was subjected to estimate the oxidative stress parameters, namely malondialdehyde (MDA) levels, superoxide dismutase (SOD) levels, and catalase (CAT) activity using the established procedure [23].
Assessment of serum biochemical parameters
The serum biochemical parameters such as alkaline phosphate (ALP), serum glutamic pyruvate transaminase (SGPT), bilirubin, and serum glutamic oxaloacetate transaminase (SGOT) were estimated as per the standard method described in the EXCEL kit leaflet (EXCEL Pvt Ltd, India) [24-26].
Histopathological studies
The thin sections of formalin fixed liver tissues were made using paraffin blocks and stained by hematoxylin and eosin stain. These stained sections were inspected under a light microscope.
Results
In vitro antioxidant activity
The antioxidant activity of LP3, LP4, and LP was evaluated by DPPH free radical scavenging activity of ferric ion reducing power assays. All the tested samples exhibited a considerable antioxidant activity compared with ascorbic acid. LP4 and LP showed significant activity when compared with LP3 (Figure 1).
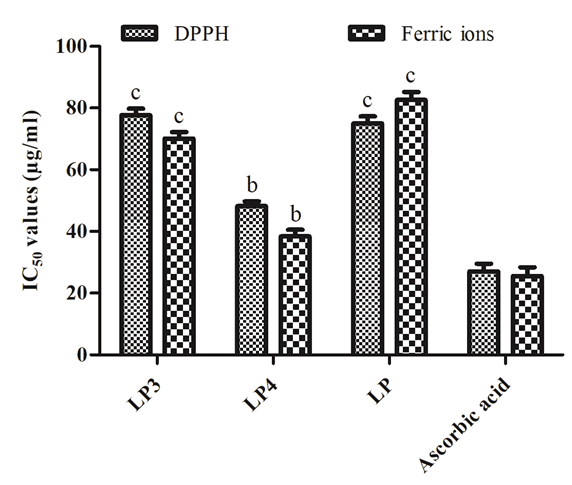
Figure 1: In vitro antioxidant activity of LP and its fractions (LP3 and LP4). Pb<0.001, Pc<0.0001, as compared with standard group using one-way ANOVA with Student-Newman-Keuls post hoc test.
Total flavonoid and phenol contents
The total flavonoid value of LP3 and LP4 was equivalent to 14.08 ± 0.40 and 18.02 ± 1.12 mg/g rutin, respectively, while the total phenolic value was equal to 100.80 ± 10.70 and 152.15 ± 12.00 mg/g of gallic acid, respectively.
Serum liver function tests
In the toxic control group, the chronic administration of ethanol (P<0.0001) significantly elevated the serum SGOT, SGPT, ALP, and bilirubin levels compared with the normal control group (Table 2). The animal group co-administered with LP3 did not show significant variation in SGPT and bilirubin levels at 100 mg/kg dose. In contrast, the rat group co-administered with LP4 offered significant dose dependent protection and significantly reduced the elevated levels of liver parameters at both low and high doses (Table 2). Moreover, co-administration of 200 mg/kg dose of LP4 (P<0.0001) offered almost equivalent protection in the liver parameters compared with silymarin (100 mg/kg) (Table 2).
Table 2: Effect of LP3 and LP4 on liver function tests.
| Groups | SGPT | SGOT | ALP | Bilirubin |
|---|---|---|---|---|
| Normal control | 35.38 ± 3.28 | 59.38 ± 3.21 | 197.55 ± 6.59 | 0.29 ± 0.30 |
| Toxic control | 74.55 ± 5.70c | 129.05 ± 6.20c | 336.38 ± 6.50c | 1.50 ± 0.63c |
| Silymarin (100 mg/kg) | 49.38 ± 2.78z | 69.88 ± 5.98z | 239.05 ± 5.11z | 0.41 ± 0.07z |
| LP3 (100 mg/kg) | 69.38 ± 5.93ns | 123.88 ± 5.51z | 320.38 ± 7.19# | 1.30 ± 0.49ns |
| LP3 (200 mg/kg) | 63.70 ± 4.49y | 107.05 ± 6.99z | 309.05 ± 8.66z | 1.25 ± 0.33x |
| LP4 (100 mg/kg) | 57.88 ± 4.00z | 78.88 ± 5.85z | 261.88 ± 8.04z | 0.57 ± 0.08z |
| LP4 (200 mg/kg) | 48.88 ± 5.75z | 65.70 ± 4.06z | 214.70 ± 9.03z | 0.60 ± 0.07z |
| All the values were expressed as mean SEM (n=6). Where, Pc<0.0001 as compared with normal control, and Px<0.05, Py<0.001, Pz<0.0001 as compared with toxic control was considered as statistically significant using one-way ANOVA with Dunnett's multiple comparison test. While P>0.05 was considered as statistically non-significant (ns) | ||||
Oxidative stress markers
The ethanol treated group (P<0.0001) significantly reduced the endogenous antioxidant enzymes, namely SOD and CAT, compared with the normal control group. The animal group co-administered with both the fractions (LP3 and LP4) offered significant protection against the ethanol-toxicity in level (Table 3). The high dose of LP3 significantly elevated the SOD and CAT levels compared with its low dose and toxic control group (Table 3). On the other hand, co-administered LP4 offered dose dependent protection, and the high dose of LP4 (P<0.0001) showed potent elevation of SOD and CAT levels similar to that of silymarin (Table 3).
Table 3: Effect of LP3 and LP4 on tissue oxidative stress markers.
| Groups | SOD | CAT | MDA |
|---|---|---|---|
| Normal control | 27.0 ± 0.33 | 58.40 ± 0.51 | 2.80 ± 0.30 |
| Toxic control | 7.8 ± 0.78c | 3.40 ± 0.44c | 12.08 ± 0.55c |
| Silymarin (100 mg/kg) | 28.68 ± 1.06z | 53.83 ± 2.01z | 2.89 ± 0.70z |
| LP3 (100 mg/kg) | 12.30 ± 0.61ns | 13.31 ± 0.50x | 9.61 ± 0.47ns |
| LP3 (200 mg/kg) | 17.93 ± 1.06y | 20.84 ± 1.03z | 6.44 ± 0.74x |
| LP4 (100 mg/kg) | 19.80 ± 2.04z | 28.18 ± 1.69z | 5.60 ± 0.44y |
| LP4 (200 mg/kg) | 26.05 ± 2.06z | 52.48 ± 1.03z | 3.44 ± 0.56z |
| All the values were expressed as mean ± SEM (n=6). Where, Pc<0.0001 as compared with normal control, and Px<0.05, Py<0.001, Pz<0.0001 as compared with toxic control was considered as statistically significant using one-way ANOVA with Dunnett's multiple comparison test. While P>0.05 was considered as statistically non-significant (ns) | |||
Further, the toxic control group (P<0.0001) significantly elevated the serum levels of MDA compared with the normal control group (Table 3). The co-administration of LP3 and LP4 significantly depleted the elevated MDA levels compared with the ethanol intoxicated group (Table 3). At 200 mg/kg dose of LP3 (P<0.05) significantly decreased the MDA levels, while 100 mg/kg of LP3 was non-significant in controlling MDA levels. LP4 markedly diminished the MDA levels in a dose dependent manner, and the effects at a higher dose (200 mg/kg) were comparable with silymarin (Table 3).
Histopathological studies
The liver’s architecture is clear, and the hepatocytes were arranged clear in normal control (Figure 2A). In contrast, the ethanol administration results in disruption of the architecture, infiltration of inflammatory cells, vacuolated hepatocytes deposition of lipids, and degenerated nuclei (Figure 2B). At a high dose, co-administration LP4 remarkably reversed the alterations induced by ethanol than LP3, supporting the research findings of biochemical (Figures 2C-2E).
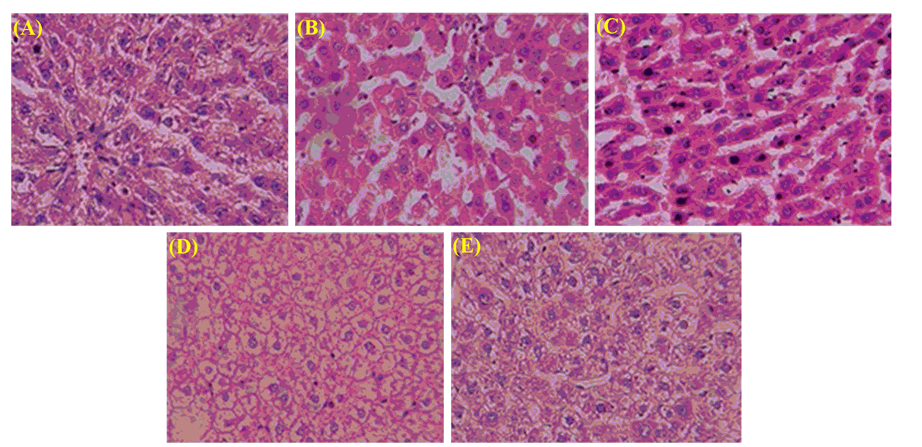
Figure 2: Histopathological studies of liver tissues of LP3- and LP4-treated groups against ethanol-induced oxidative stress. (A) normal control (0.2 ml, vehicle); (B) toxic control (ethanol, 5 g/kg, 20%); (C) standard (silymarin (100 mg/kg)+ethanol (5 g/kg, 20%)); (D) LP3 (200 mg/kg)+ethanol (5 g/kg, 20%); (E) LP4 (200 mg/kg)+ethanol (5 g/kg, 20%).
Discussion
Liver fibrosis is the combined outcome of both sustained inflammatory and wound healing processes of repeated injury. Normally, once the injury occurs, the parenchyma cells will be regenerated, repair the injured region associated with inflammation. Repeated injury leads to sustained inflammation and wound healing process leading to fibrosis and cirrhosis [1-5]. The alcohol is a well-known chemical toxicant that induces liver fibrosis. The alcohol induced liver fibrosis model in animals (rat/mice) has been used by various investigators to study different aspects of liver fibrosis and healing [5,26]. The repeated administration of the alcohol induces lipid peroxidation and injured hepatocytes release the cellular components [26].
Since ancient times, lichens parts are being used to treat hepatotoxicity both in Ayurveda and Unani systems [10,26- 28]. Based on this evidence, the search for new lead molecules for liver disorders from natural sources is a crucial point of interest. As a result, many lichens and their phytochemicals were tested for antioxidant and hepatoprotective potentialities [26-31]. Therefore, in the present study, L. pulmonaria were extracted with ethanol and fractionated. The two obtained fractions (LP3 and LP4) exhibited significant DPPH free radical scavenging activity and ferric ion reducing power capabilities attributed to the substantial quantity of phenol and flavonoid compounds [32,33] (Figure 1).
As mentioned earlier, acetaldehyde accumulation in the liver inhibits the transport pumps present on the hepatocytes’ membrane, thereby outflow the liver enzymes such as SGOT, SGPT, ALP, and bilirubin into serum results in hepatotoxicity [32-34]. In the current investigation, similar observations were noticed in the toxic control group. Co-administration of LP3 and LP4 significantly reduced the elevated levels of SGOT, SGPT, ALP, and bilirubin (Table 2). Notably, co-administration of a higher dose of LP4 displayed equivalent protection in the liver parameters compared with silymarin (Table 2).
Generally, oxidative stress is the pathogenesis of numerous chronic diseases that affects the imbalance between the antioxidants and reactive oxygen species [32]. The endogenous antioxidant enzymes like SOD and CAT play a vital role in catalyzing the H2O2 into the water, the major precursor of ROS. The depletion of SOD and CAT results in oxidative stress in surrounding tissues and causes tissue and organ necrosis [35]. Moreover, the excess generation of ROS plays a vital role in hepatotoxicity’s etiology [36]. The chronic administration of ethanol activates the kuffer cells, thereby generates ROS and pro-inflammatory substances [37]. These excess ROS covalently binds to the hepatocytes’ membrane and results in oxidation of polyunsaturated lipids that markedly elevate the MDA levels [38]. Further, excess accumulation of both ROS and MDA levels together causes oxidative stress in hepatocytes [39].
The present study’s findings indicate that ethanol administration markedly elevated the MDA levels by depleting the endogenous antioxidants (SOD and CAT) (Table 3). In contrast, the administration of fractions significantly antagonized the oxidative stress induced by ethanol by enhancing the SOD and CAT levels and simultaneously depleting the MDA levels in a dose dependent manner when comparing with toxic control (Table 3). The co-administration of 200 mg/kg of LP4 exhibited marked protection of tissue oxidative stress markers similar to silymarin. Moreover, our outcomes are identical to the previous study models of hepatotoxicity [40,41].
The histopathological studies are in support of the hepatoprotective activity of LP3 and LP4. The chronic administration of alcohol causes disruption of hepatocytes, infiltration of cells, and ballooning of cells [28], whereas both fractions’ co-administration reversed the damage effects caused by alcohol. Among both, LP4 maintained the better structural integrity of hepatocytes and antagonized ethanol induced toxicity than LP3. These histopathological results support the hepatoprotective potentiality of L. pulmonaria against ethanol-intoxicated rats.
Conclusion
To conclude, the present study showed that fractions of L. pulmonaria have a good amount of total flavonoid and phenolic contents with potent antioxidant activity. Additionally, LP3 and LP4 unexpectedly improved serum liver parameters and oxidative stress markers in rats induced with ethanol, which is attributed to viz., radical scavenging and antioxidant activities. Also, the co-administration of these fractions showed hepatoprotective effects in ethanol induced Wistar albino rats. Hence, L. pulmonaria could be used in the treatment of different liver ailments.
Conflict of Interest
The authors have no conflicts of interest regarding this investigation.
Acknowledgments
None.
References
- H. Polimati, R.P. Pragada, N.H. Thuan, V.B. Tatipamula, Hepatoprotective potential of bioflavonoids, Stud Nat Prod Chem, 72 (2022), 259-285.
- S.K. Asrani, H. Devarbhavi, J. Eaton, P.S. Kamath, Burden of liver diseases in the world, J Hepatol, 70 (2019), 151-171.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- L.A. John, N. Kannappan, P. Manojkumar, Evaluation of hepatoprotective activity in methanolic extract of aerial parts of Hibiscus surattensis, Res J Pharm Technol, 13 (2020), 4635-4640.
- T.B. Nguyen, D.N. Do, T. Nguyen-Thanh, V.B. Tatipamula, H.T. Nguyen, Identification of five hub genes as key prognostic biomarkers in liver cancer via integrated bioinformatics analysis, Biology, 10 (2021), 957.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- H. Zhao, N. Cheng, L. He, G. Peng, X. Xue, et al., Antioxidant and hepatoprotective effects of A. cerana honey against acute alcohol-induced liver damage in mice, Food Res Int, 101 (2017), 35-44.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- H.T. Nguyen, T.Y. Vu, T.C. Dakal, B. Dhabhai, X.H. Nguyen, et al., Cleroda-4 (18), 13-dien-15, 16-olide as novel xanthine oxidase inhibitors: An integrated in silico and in vitro study, PloS One, 16 (2021), e0253572.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- L.E. Nagy, Molecular aspects of alcohol metabolism: Transcription factors involved in early ethanol-induced liver injury, Annu Rev Nutr, 24 (2004), 55-78.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, K.N. Killari, H. Polimati, A. Ketha, Chemical and biological investigations on mangrove associated lichens (manglicolous lichens) from India: A review, Indian J Pharm Sci, 83 (2021), 186-194.
- O.R. Koch, G. Pani, S. Borrello, R. Colavitti, A. Cravero, et al., Oxidative stress and antioxidant defenses in ethanol-induced cell injury, Mol Asp Med, 25 (2004), 191-198.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- S.D. Crawford, Lichens used in traditional medicine, Lichen secondary metabolites- Springer, (2019), 31-97.
- H.T. Nguyen, T.Y. Vu, V. Chandi, H. Polimati, V.B. Tatipamula, Dual COX and 5-LOX inhibition by clerodane diterpenes from seeds of Polyalthia longifolia (Sonn.) Thwaites, Sci Rep, 10 (2020), 15965.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, G.S. Vedula, Fibrinolytic, anti-inflammatory and cytotoxic potentialities of extracts and chemical constituents of manglicolous lichen, Graphis ajarekarii Patw. & C. R. Kulk, Nat Prod J, 10 (2020), 87-93.
- T.T. Nguyen, S. Nallapaty, G.K. Rao, S.T. Koneru, S.S.P. Annam, et al., Evaluating the in vitro activity of depsidones from Usnea subfloridana Stirton as key enzymes involved in inflammation and gout, Pharm Sci, 27 (2020), 291-296.
- V.B. Tatipamula, G.S. Vedula, A.V.S. Sastry, Antarvedisides A-B from manglicolous lichen Dirinaria consimilis (Stirton) and their pharmacological profile, Asian J Chem, 31 (2019), 805-812.
- V.B. Tatipamula, G.S. Vedula, A.V.S. Sastry, Chemical and pharmacological evaluation of manglicolous lichen Roccella montagnei Bel em. D. D. Awasthi, Future J Pharm Sci, 5 (2019), 8.
- P. Haritha, S.K. Patnaik, V.B. Tatipamula, Chemical and pharmacological evaluation of manglicolous lichen Graphis ajarekarii Patw. & C. R. Kulk, Vietnam J Sci Technol, 53 (2019), 300-308.
- K. Venkatesham, D.C. Babu, V.B. Tatipamula, R.A. Bunce, C.B. Rao, et al., Synthesis of n-alkyl terminal halohydrin esters from acid halides and cyclic ethers or thioethers under solvent-and catalyst-free conditions, RSC Adv, 4 (2014), 51991-51994.
- V.B. Tatipamula, K.N. Killari, K.V. Gopaiah, A. Ketha, GC-MS analysis of ethanol extract of Taxithelium napalense (Schwaerg) Broth along with its alpha-glucosidase inhibitory activity, Indian J Pharm Sci, 81 (2019), 569-574.
- H.T. Nguyen, T.Y. Vu, A.V. Kumar, V.N. Hoang, P.T. My, et al., N-Aryl iminochromenes inhibit cyclooxygenase enzymes via π-π stacking interactions and present a novel class of anti-inflammatory drugs, RSC Adv, 11 (2021), 29385-29393.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, S.S.P. Annam, H.T. Nguyen, H. Polimati, R.P. Yejella, Sekikaic acid modulates pancreatic β-cells in streptozotocin-induced type 2 diabetic rats by inhibiting digestive enzymes, Nat Prod Res, 35 (2021), 5420-5424.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, M.K. Kolli, S.B. Lagu, K.R. Paidi, P.R. Reddy, R.P. Yejella, Novel indolizine derivatives lowers blood glucose levels in streptozotocin-induced diabetic rats: A histopathological approach, Pharmacol Rep, 71 (2019), 233-242.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, A. Ketha, S. Nallapaty, H. Kottana, S.T. Koneru, Moss Octoblepharum albidum Hedw: Isolation, characterization, in vitro and in vivo antidiabetic activities, Adv Trad Med, 2 (2021), 351-360.
- V.B. Tatipamula, B. Kukavica, Protective effects of extracts of lichen Dirinaria consimilis (Stirton) D.D. Awasthi in bifenthrin- and diazinon-induced oxidative stress in rat erythrocytes in vitro, Drug Chem Toxicol, 45 (2020), 680-687.
[Crossref] [Google Scholar] [PubMed]
- S. Prakash, A.K. Verma, Effect of arsenic on serum biochemical parameters of a fresh water cat fish, Mystus vittatus, Int J Biol Innov, 2 (2020), 11-19.
- S. Kumar, R.P. Raman, K.P. Prasad, P.P. Srivastava, S. Kumar, et al., Effects on haematological and serum biochemical parameters of pangasianodon hypophthalmus to an experimental infection of Thaparocleidus sp. (Monogenea: Dactylogyridae), Exp Parasitol, 188 (2018), 1-7.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- P. Lodhi, N. Tandan, N. Singh, D. Kumar, M. Kumar, Camellia sinensis (L.) kuntze extract ameliorates chronic ethanol-induced hepatotoxicity in albino rats, Evid Based Complement AlterN Med, 2014 (2014), 787153.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, S.S.P. Annam, Antimycobacterial activity of acetone extract and isolated metabolites from folklore medicinal lichen Usnea laevis Nyl. against drug-sensitive and multidrug-resistant tuberculosis strains, J Ethnopharmacol, 282 (2022), 114641.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- N.H. Thuan, A. Shrestha, N.T. Trung, V.B. Tatipamula, D.V. Cuong, et al., Advances in biochemistry and the biotechnological production of taxifolin and its derivatives, Biotechnol Appl Biochem, 69 (2021), 848-861.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- K.R. Paidi, V.B. Tatipamula, M.K. Kolli, S.S.P. Annam, V.R. Pedakotla. Synthesis of imidazo [1, 2-b] pyridazine comprised piperazine, morpholine derivatives as potent antimycobacterial agents with in vivo locomotor activity, Anti-Infective Agents, 15 (2017), 131-139.
- V.B. Tatipamula, H. Polimati, K.V. Gopaiah, A.K. Babu, S. Vantaku, et al., Bioactive metabolites from manglicolous lichen Ramalina leiodea (Nyl.) Nyl, Indian J Pharm Sci, 82 (2020), 379-384.
- T.T. Nguyen, A. Ketha, H.V. Hieu, V.B. Tatipamula, In vitro antimycobacterial studies of flavonols from Bauhinia vahlii Wight and Arn, 3 Biotech, 11 (2021), 128.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, B. Kukavica, Phenolic compounds as antidiabetic, anti‐inflammatory, and anticancer agents and improvement of their bioavailability by liposomes, Cell Biochem Funct, 39 (2021), 926-944.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- N.H. Thuan, H. Polimati, R. Alluri, V.B. Tatipamula, Bioassay-guided isolation of antimycobacterial substances from the traditionally used lichen Cladonia pyxidata (L.) Hoffm, 3 Biotech, 12 (2022), 95.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- N.H. Thuan, V.B. Tatipamula, T.T. Viet, N.Q.D.T. Tien, N.H. Loc, Bioproduction of eriodictyol by Escherichia coli engineered co-culture, World J Microbiol Biotechnol, 38 (2022), 112.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, A. Ketha, Manglicolous lichen Parmotrema tinctorum (Despr. ex Nyl.) Hale: Isolation, characterization and biological evaluation, Indian J Chem Sect B, 59 (2020), 856-861.
- V.B. Tatipamula, K.N. Killari, K. Prasad, G.S. Rao, M.R. Talluri et al., Cytotoxicity studies of the chemical constituents from marine algae Chara baltica, Indian J Pharm Sci, 81 (2019), 815-823.
- S.A. Abdellatief, A.A.A. Galal, S.M. Farouk, M.M. Abdel-Daim, Ameliorative effect of parsley oil on cisplatin-induced hepato-cardiotoxicity: A biochemical, histopathological, and immunohistochemical study, Biomed Pharmacother, 86 (2017), 482-491.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- S.L. Yan, Z.H. Wang, H.F. Yen, Y.J. Lee, M.C. Yin, Reversal of ethanol-induced hepatotoxicity by cinnamic and syringic acids in mice, Food Chem Toxicol, 98 (2016), 119-126.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- V.B. Tatipamula, V.K. Tatipamula, Dibenzofurans from Cladonia corniculata Ahti and Kashiw inhibit key enzymes involved in inflammation and gout: An in vitro approach, Indian J Chem Sect B, 60 (2021), 715-719.
- N. Ilaiyaraja, F. Khanum, Amelioration of alcohol-induced hepatotoxicity and oxidative stress in rats by Acorus calamus, J Diet Suppl, 28 (2011), 331-345.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
- M.A. Alsaif, Effect of thymoquinone on ethanol-induced hepatotoxicity in Wistar rats , J Med Sci, 7 (2007), 1164-1170.
[Crossref] [Google Scholar] [PubMed] [Research Gate]
Copyright: © 2022 Kishore Naidu Killari, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

