Research Article: Journal of Drug and Alcohol Research (2025) Volume 14, Issue 12
Last Information on Gait Apraxia, Diagnostic Procedures and Systematic Review
Lourdes de Fatima Ibanez Valdes1 and Humberto Foyaca Sibat2*2Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa
Humberto Foyaca Sibat, Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa, Email: humbertofoyacasibat@gmail.com
Received: 19-Nov-2025, Manuscript No. JDAR-26-176231; Editor assigned: 21-Nov-2025, Pre QC No. JDAR-26-176231 (PQ); Reviewed: 05-Dec-2025, QC No. JDAR-26-176231; Revised: 22-Jan-2026, Manuscript No. JDAR-26-176231 (R); Published: 29-Dec-2025, DOI: 10.4303/JDAR/236487
Abstract
Introduction: Apraxia (motor agnosia) is a neurological condition characterized by the desire and ability to move which is impeded by the inability to performed it due to corpus callosum damage (callosotomy/ anterior cerebral artery stroke/mass), left inferior parietal lobule, the premotor cortex, supplementary motor area and convexity lesions affecting sequencing of purposeful, the planning and to perform skilled movements, including dressing, speaking or using tools, no related to motor disorder.
Objectives: The main aim of this study is to discuss the last information released into the medical literature regarding to Gait Apraxia (GA) and the novel diagnostic procedure reported in the available medical literature.
Methods: A wide searching of the medical literature of the following database PubMed/MEDLINE, Scopus and Embase databases was performed to find articles related to novel information on GA and diagnostic procedures. From 01st January 1989 to 30th November 2025, we searched the medical literature, following the PRISMA guidelines. The authors searched the scientific databases, Scopus, Embassy, Medline, and PubMed Central using the following searches: “gait apraxia” OR “walking skills” OR “apraxia of postural transitions” OR “diagnostic tools” OR “limb apraxia”, OR “pathophysiology of apraxia” OR “treatment/management of apraxia” OR “corticobasal syndrome” OR “normal pressure hydrocephalus”.
Results: Searching literature retrieved 359 articles. 123 duplicated studies and after reviewing the titles and abstracts were removed, 89 publications were selected. After applying the inclusion/exclusion criteria 34 articles were excluded; therefore, 12 studies investigated the role of diagnostic procedures in the assessment of GA were selected.
Conclusions: The GA in patients presenting iNPH is characterized by decreased flexion at the hip joint, a decreased toe‐off angle, heel strike angle, associated shorter strides, diminished speed, and prolonged time for swing and single support.
Keywords
Gait apraxia; Assessment of walking skills; Apraxia of postural transitions; Neuropsychological disorder; Diagnostic tools
Introduction
Apraxia (motor agnosia) is a neurological condition characterized by the desire and ability to move which is impeded by the inability to performed it due to corpus callosum damage (callosotomy/anterior cerebral artery stroke/mass), left inferior parietal lobule, the premotor cortex, supplementary motor area and convexity lesions affecting sequencing of purposeful, the planning and to perform skilled movements, including dressing, speaking or using tools, no related to motor disorder. Apraxia is mainly divided into two subtypes: The inability to perform learned movements on command (ideomotor) and the incapacity to conceptualise and sequence a series of movements (ideational). Apraxia can also be divided into loss of dexterity (limb-kinetic Apraxia), task-specific apraxia (such as gait or dressing apraxia) and speech/ language disorder not related to dysarthria/aphasia (verbal apraxia). Other authors described apraxia as “a disorder of skilled movement not caused by weakness, akinesia, deafferentation, abnormal tone or posture, movement disorders such as tremors or chorea, intellectual deterioration, poor comprehension, or uncooperativeness.”
The most typical type of limb apraxia, composed of ideational, ideomotor and limb-kinetic subtypes and each one is related to a lesion in specific cerebral areas involving the basal nuclei, the fronto-parietal cortex and the white matter fasciculi [1,2].
Other investigators documented that apraxia is a neurological disorder that affects motor activities, cognition, planning and task performance, with normal motor pathways, sensory systems, cerebellar coordination and cognition as potential aetiologies of these deficits. They also documented that some aetiologies, such as neurocognitive disorders, brain injuries, dementia, space-occupying lesions and stroke, can be identified and recommended to manage this disorder by treating the underlying disease and providing supportive procedures. These authors described the clinical features of apraxia in different group of patients and different scenarios, they also identified the particular capacities and needs in apraxia cases, choice the most accurate assisting device and necessary technology to provide the best possible assistance to patients with apraxia and included other medical disciplines like speech-language pathologists and occupational therapists among other in the therapeutic management of cases with apraxia [3].
The medical term praxis refers to the ability to perform skilled actions and involves positive (activation) or negative (inhibition) electrical impulses in cortical activity. Dysfunctional praxis is diagnosed as apraxia, which can be confirmed by performing a comprehensive assessment of patients that includes a detailed history of the present complaint, a general neurological examination and apraxiaspecific testing.
Ideational apraxia refers to the loss of the capacity to perform a sequence of movements, whereas the loss of the concept itself is called conceptual apraxia.
The approach to the management of apraxia includes addressing its underlying aetiology through occupational, physical or other task-specific therapeutic interventions, followed by counselling. The extent of the intervention is directly related to the type of apraxia and its impact on the patient’s daily activities.
Sometimes, some clinical disturbances associated with apraxia, such as dysgraphia, dyscalculia, social anxiety, low self-esteem, dysphasia or loss of awareness, can be observed.
The chance to acquire a particular type of apraxia is related to the chance of a particular cause. In other words, different types of apraxia depend on the risk of developing its associated aetiology.
The prevalence of apraxia in cases presenting stroke in the left cerebral hemisphere is around 50% to 80% while cases with stroke in the right cerebral hemisphere have a prevalence of 30% to 50%.
Patients with dementia syndrome have a 90% prevalence, 25% with multiple sclerosis and 19% to 45% in those cases with traumatic brain injuries [3].
Unfortunately, the inability to carry out learning skills in patients with apraxia profoundly affects their capacity to conduct personal activities of daily living and maintain their proper independence. Some necessary protective measures must be taken to avoid domestic accidents, outside injuries and unnecessary risks. Obviously, the prognosis depends on the underlying condition.
Notwithstanding, that incapacity to perform skilled movements leads to depression, personal frustration, decreased motivation and deterioration of the usual daily activities, which require sustainable communication with caregivers, special counselling of family/relative members to reach understanding care goals, realistic expectations and a better recovery journey. All people involved in the rehabilitation process, including caregivers, patients, families, relatives and other interested parties, must actively work toward this goal. The active involvement of professional nurses, occupational therapists, physiotherapists, family members, physiatrists, psychiatrists, primary care physicians, radiologists, neurologists, counsellors and social workers is paramount in managing these cases to enhance patient outcomes. Psychological intervention across the recovery process and rehabilitation program is vital for long-term patient well-being. However, early diagnosis and prompt interprofessional management of the apraxia can improve the prognosis.
The medical terminology for apraxia must exclude akinesia, muscle weakness, abnormal muscle tone or posture, sensory disturbance, decreased comprehension, movement disorders such as tremor or chorea, cognitive decline or lack of cooperation [4]. Nevertheless, apraxia is most seen in patients presenting a variety of neurological and psychiatric conditions [5-9].
In around 50% of patients presenting with left brain stroke, limb apraxia can be present.
Limb apraxia is a condition related to lesions affecting the ventro-dorsal pathways at the intersection of cognition and motor control [10].
Recently, Igari and collaborators from the Department of Neurology, University of Tsukuba, Japan, reported two cases presenting decreased blood flow around the Supplementary Motor Area (SMA) in Corticobasal Syndrome (CBS), characterised by incapacity of start and coordinating walking despite normal conceptual movement abilities, in whom lower limb apraxia was ruled out. They documented the associated CBS as a clinical phenotype of corticobasal degeneration, which upper-limb apraxia usually manifests, whereas lower-limb or gait apraxia is usually absent. However, these authors confirmed GA due to SMA dysfunction by Single-Photon Emission Computed Tomography (SPECT) [11].
Recently, Balash et al. reported, for the first time, Limb-Kinetic Apraxia of the Legs (LK-L) in patients with Parkinson’s Disease (PD). They delivered a new approach to LK-L in PD cases, focusing on its rigidity and bradykinesia and their influence on patients’ quality of life. These authors also hypothesised that, because bipedal reciprocal coordination requires high concentration and attention, a variety of differences in the quality and rate of execution of this test should be observed, reflecting the prominent role of apraxia in the dynamic planning of motor activities in PD patients [12].
The main aim of this study is to discuss the last information released from the medical literature regarding to Gait Apraxia (GA) and the most confident diagnostic procedure reported.
Materials and Methods
A wide searching of the medical literature of the following database PubMed/MEDLINE, Scopus and Embase databases was performed to find articles related to novel information on GA and diagnostic procedures.
From 01st January 1989 to 30th November 2025, we searched the medical literature, following the PRISMA guidelines. The authors searched the scientific databases, Scopus, Embassy, Medline and PubMed Central using the following searches: “gait apraxia” OR “walking skills” OR “apraxia of postural transitions” OR “diagnostic tools” OR “limb apraxia”, OR “pathophysiology of apraxia” OR “treatment/Management of apraxia” OR “corticobasal syndrome” OR “normal pressure hydrocephalus”.
The systematic review performed in this study followed the guidelines recommended by PRISMA (2020 statement).
Search strategy
A systematic search was done through the following databases: PubMedCentral/MEDLINE, Scopus and Embase to identify publications related to novel information related to pathophysiology of GA, management and diagnostic procedures.
Only articles published in English, Spanish and Portuguese were selected. Editorials, preclinical studies and conference proceedings were excluded.
Selection of study
Both investigators (LdeFIV. and HFS) separately reviewed the abstracts and titles of the selected publications and independently reviewed the full-text versions of the identified articles to determine their eligibility for inclusion. Medical investigations delivering information on diagnostic procedure to assess GA were selected for inclusion in this systematic study. Furthermore, publications without a clear protocol of confirmatory diagnosis, papers with lack of analysis, publications with incomplete data or without mention exact number of patients were not considered for inclusion.
Selection criteria
The following criteria were included: Articles with detailed pathogenesis and/or drug management. Clinical features of LIS, and demographic information.
Exclusion criteria were applied:
• Inaccessibility to full text.
• Articles with unclear pathogenesis.
• Lack of relevant clinicopathological data.
• Non-original studies (i.e., editorials, letters, conference proceedings, book chapters).
• Animal model studies; and (6) non-/Spanish/ Portuguese/English studies.
Data extraction and quality assessment
The studies’ quality assessment including applicability concerns and risk of bias was categorized as good, poor, fair or reasonable, in agreement with the National Institutes of Health criteria and the Quality Assessment of Diagnostic Accuracy Studies version 2 (QUADAS-2) evaluation. Quality evaluation was made separately by both authors and disagreements were resolved by scientific discussion and final agreement.
Data collection, extraction and bias assessment
All abstracts and titles with the inclusion criteria were revised by two before cited authors to collect relevant information for the review. For each publication selected in the review, data concerning to author’s name, age, year of publication, country of information’ source, type of the study, radiopharmaceutical employed, total of cases included and most probable aetiology of the GA, were collected. The selected data from eligible publications were introduced into an updated Excel software program.
Outcome measures
Our plan aimed to select the most relevant publications relating to the clinical benefit and real-world effectiveness of different diagnostic procedures for GA. This investigation also identified safety outcomes data, including adverse and serious adverse events across the clinical investigations.
Statistical analysis
Statistical investigation was done through XLSTAT (addon for Microsoft Excel, version 2021.4.1, Addinsoft SARL) and RStudio (version 4.3.1, https://www.rstudio.com/).
Results and Discussion
Literature search
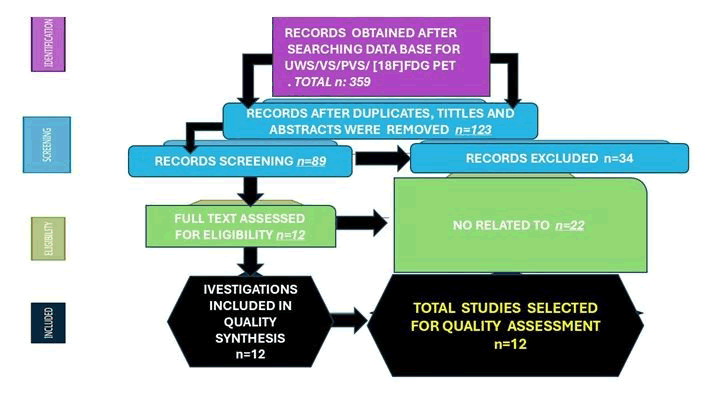
Searching literature retrieved 359 articles. 123 duplicated studies and after reviewing the titles and abstracts were removed, 89 publications were selected. After applying the inclusion/exclusion criteria 34 articles were excluded; therefore, 12 studies investigated the role of diagnostic procedures in the assessment of GA were selected (Figure 1).
Figure 1: PRISMA flow diagram with included publications
To provide an accurate management of this searching, the authors used a QUADAS-2 evaluation to determine low/moderate risk of bias for almost all publications. The diagnosis of GA was made in overall patients according to current proposed criteria. Authors considered the elevated technical differences obtained from quite different diagnostic protocols performed in several studies.
Noticeably, in some publications, small and mixed cohorts were processed including different types of diagnostic procedures leading to in fewer GA cases examined under the same protocol.
Comments and final remarks
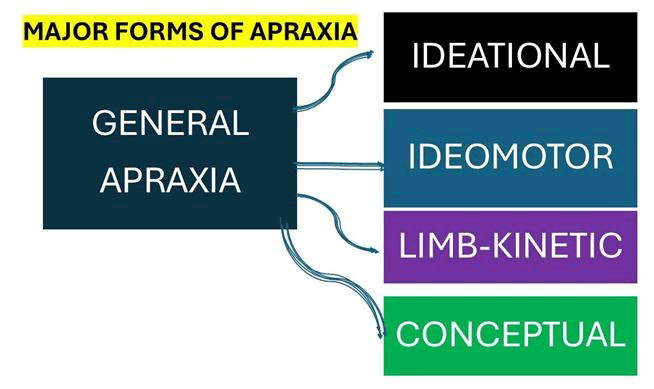
Different types of apraxia were described before according to their clinical features. Here, we listed the most common types of major forms of apraxia in Figure 2 for general apraxia.
Figure 2: Shows different types of general apraxia: Ideomotor apraxia: Patients exhibit an inability to pantomime or imitate gestures. Moreover, they experience challenges with spatiotemporal orientation and positioning. Movements involving their fingers, hands, and arms display abnormal trajectories. Ideational apraxia: Patients struggle to identify the correct sequence of actions necessary to accomplish a task. Even when provided with a list of required steps, they may encounter difficulties organizing them accurately. Limb-kinetic apraxia: Patients exhibit incorrect fine motor actions, particularly with their hands, when attempting to perform a learned task. For instance, they may struggle with rotating a coin using their thumb, index finger, and middle finger. Conceptual apraxia: Patients face challenges identifying the appropriate tool for a given task. When presented with a tool, they may struggle to discern its purpose. Additionally, they may have trouble in pantomiming the correct utilization of a tool
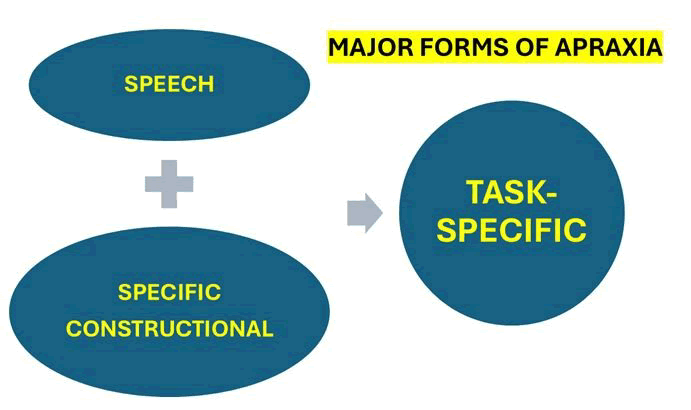
The task specific types of general apraxia which are represented in Figure 3.
Figure 3: The task specific types of general apraxia
Brief comment on the pathophysiology of apraxia
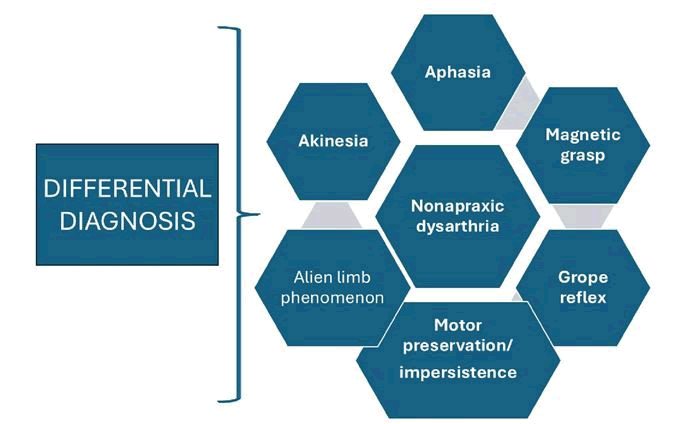
Before discussing the relationship between apraxia and the anatomical components involved in its pathophysiology, we listed the main pathological conditions to be considered in the differential diagnosis of Apraxia as is shown in Figure 4.
Figure 4: The differential diagnosis of apraxia
The first comment is to highlight the role played by frontal motor areas and the left inferior parietal lobule in praxis and apraxia. The second one concerns remarking the role of auditory, verbal, visual or tactile stimulation in the activation mechanism of the praxis network through direct stimulation of cortical neurons in the left parietal lobe, which trigger the praxis mechanism. The praxis network comprises other anatomical regions, such as prefrontal, temporal, thalamic and basal ganglia regions, as well as connecting white matter. Therefore, selected injuries in these areas can cause Apraxia according to the role of the affected structure. To assess praxis, two different activities should be considered:
• The ones recalled from the memory system.
• The ones are imitated directly.
These two activities can be separated into three types: Transitive gestures, intransitive gestures and pantomime of tool use. Transitive gestures involve an object, such as flipping a coin or swinging a hammer. The next one, also known as symbolic gestures (intransitive gestures), involves performing a greeting salute or waving goodbye without depending on an object. The last one (pantomime) mimics or simulates the use of an instrument, tools or other objects without their presence.
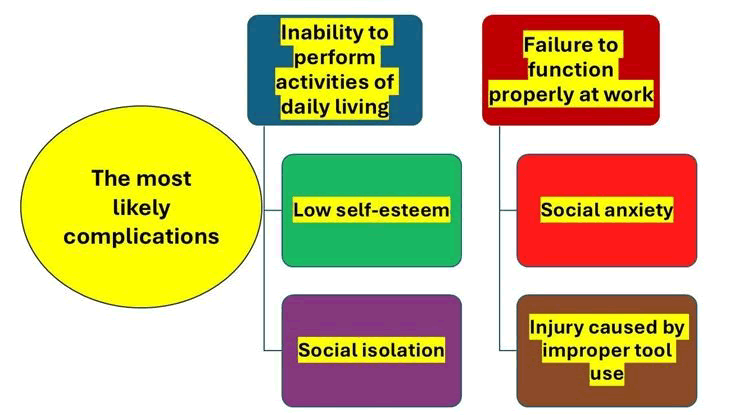
The most typical complications of apraxia are represented in Figure 5.
Figure 5: The most typical complications of apraxia
Imaging (MRI/PET) and laboratory investigations can be used to identify aetiologies associated with apraxia, such as stroke, TBI, multiple sclerosis and neurodegenerative conditions.
Brief comments on apraxia assessment and therapy
The main aim of therapeutic management of apraxia is to initiate a rehabilitation program, including OT, speech therapy and physiotherapy, as well as managing the underlying condition. Currently, there is no available drug therapy to treat patients with apraxia. Besides medical treatment for sleep apnea, atrial fibrillation, hypertension, diabetes mellitus, tobacco use and alcohol use disorder are underlying problems.
However, transcranial magnetic stimulation may yield better outcomes for these patients [3]. Disturbance of pantomime tasks or gesture imitation has been documented in neurological conditions such as Parkinson’s disease [13], Alzheimer’s disease [14], corticobasal syndrome [15], multiple sclerosis [16] and Richardson’s syndrome [17], plus psychiatric diseases like bipolar disorder (type 1) [18], major depressive disorder [19] and schizophrenia [20].
Some investigators have suggested brain stimulation techniques as part of the therapy of limb apraxia, considering that it may strengthen training effects or virtual and augmented surrounding environment, offering better compensating planning and a safe opportunity to create more complex training reality by also introducing threedimensional holographic cues with the intention to improve the pantomime of tool use, compared to two-dimensional cues [21].
Other good therapeutic outcomes in the management of ideomotor apraxia can be achieved by using virtual reality appropriately [22].
Obviously, given the ongoing discovery of novel technologies, a sustained reassessment of current diagnostic and treatment procedures is necessary. In cases presenting aphasia due to ischemic stroke on the proper inferior parietal cortex, stimulation techniques can be administered. For example, administering transcranial continuous theta burst stimulation (inhibitory approach) may diminish the inhibitory effect at the same level on the contralateral cerebral hemisphere in schizophrenic patients [23] and ischemic stroke in the left cerebral hemisphere [24].
On the other hand, the excitatory stimulation by Transcranial Magnetic Stimulation (TMS) of the left lower frontal circumvolution can improve apraxia in patients with schizophrenia [23].
Brief comments on Gait Ataxia (GA)
Meyer and Barron described GA as “the loss of ability to use the lower limbs in the act of walking properly” [25] Della Sala, from the Department of Psychology, University of Aberdeen, UK and his collaborators reported a 64-year-old male patient presenting with difficulties in standing from a chair or sitting on command a remarkable impediment to lying supine on the examining bed and the inability to roll over. His partner said he needed her support to get into and out of bed. The way of walking was hesitant, with perseverance of individual components of a movement sequence. Patient was fully aware of his limitations, never complained about psychiatric, cerebellar, extrapyramidal or cognitive problems. An aetiological diagnosis of a bilateral ischemic infarct in the anterior cerebral artery territory, affecting the supplementary motor areas of both cerebral hemispheres, leading to Gait Apraxia (GA), was confirmed. These investigators identified 49 patients with similar clinical features in the same affected regions. They documented the differential diagnosis between GA and other conditions related to pyramidal lesions or sensorial deficits, ataxia (cerebellar, vestibular or proprioceptive), old age (for example, bone and joint degeneration, muscle wasting or arteriosclerosis obliterans of the lower limbs), dystonia or dyskinesias (due to basal ganglia disorders), among others [25].
The same authors describe GA as a disorder characterised by episodes of freezing, short shufling steps, limitations in initiation of walking, impaired balance and difficulties in postural transitions, after ruling out other potential aetiologies [26]. The first aspect to differentiate GA from lower limb apraxia is based on the patient’s capacity to perform actions [27].
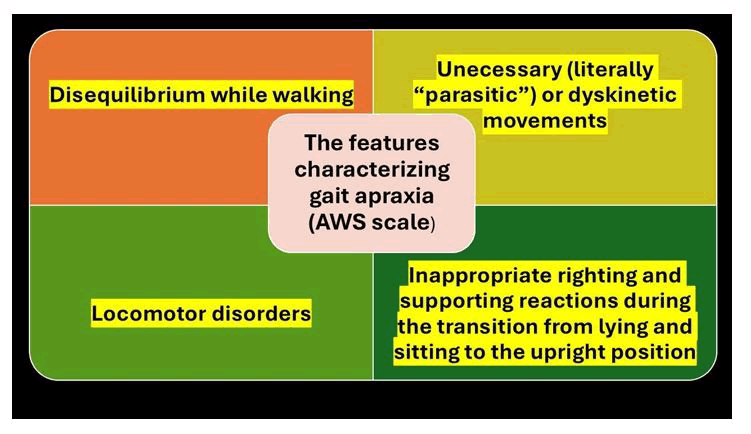
In Figure 6 are illustrated the main features characterizing gait apraxia.
Figure 6: The main features characterizing gait apraxia
Apraxia is characterised by a combination of signs, including locomotor abnormalities, inappropriate postural responses, loss of balance and equilibrium and delayed gait ignition.
Stance, trunk movements and gait are combined and practised in a remarkable way. Associated dysfunctional movements to GA include disturbances of trunk motility, such as difficulties with rising from sitting, lying down, sitting down, rolling over in bed or turning around while standing [27]. On the other hand, stance disabilities include disturbance of balance during walking, turning to any direction, standing in an upright position and falling due to loss of anticipatory and reactive postural responses, frozen foot, stepping, shufling, leg hypokinesia, obstructive leg crossing, counterproductive, bizarre and perseverative leg movements besides to lower limbs hypotonia, dyskinesias, dystonia and transient walking arrest like frontal inertia. The most typical lesion in GA is in the precentral regions of the frontal lobes [27].
GA disorder has been called as “trepidante abasie”, “bewegungstechnische schwierigkeit” (loss of the technical requirements of the movements) “Torso apraxie” (trunk apraxia), “déséquilibration frontale” (frontal imbalance), gait ignition failure, “eingentumliche Ungeschicklichkeit” (remarkable clumsiness), “Tapsigkeit” (gawky movements), “Unbeholfenheit” (awkwardness), “corticale apraxie”, slipping clutch syndrome, lower half parkinsonism, “frontale ataxie” (frontal ataxia), melokinetic apraxia, higher level gait disorder, “gliedkinetische apraxie” (limbkinetic apraxia), “motorische apraxie”, “innervatorische apraxie” (innervatory apraxia), “bilaterale motorische apraxie”, and frontal gait disorder [25].
Brief comment on corticobasal syndrome
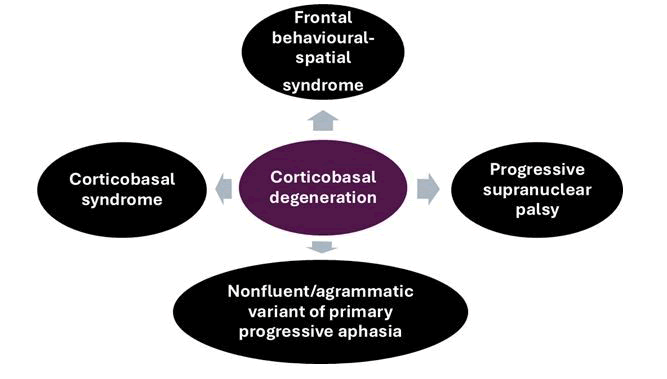
Cortico-Basal Syndrome (CBS), as one of the presentations of cortico-basal degeneration. A variety of CBS clinical phenotypes are represented in Figure 7.
Figure 7: A variety of CBS clinical phenotypes
The clinical features of CBS (progressive neurodegenerative disorder) are listed as asymmetric limb apraxia and akinetic rigidity involving the basal ganglia and cortical dysfunction, with the upper limb apraxia being the hallmark. However, lower-limb apraxia has been reported, whereas gait apraxia is underdiagnosed in these cases.
Brief comments on GA diagnostic tools
As previously cited, GA is a neurological condition that impairs voluntary and purposeful movements of the lower extremities, causing an inability to walk properly without evidence of corticospinal tract damage, ataxia, vestibular lesions or cognitive decline. Unfortunately, the lack of standardised diagnostic tools for GA in diagnostic procedures is quite common. However, an adapted Italian “Assessment of Walking Skills” (AWS) scale from the English version was initially used to evaluate trunk and walking movements and confirm GA in people presenting with Alzheimer’s Disease (AD). Now, a new method has been introduced to assess trunk and leg movements relative to a healthy control group, with a scoring range of 0-42. This procedure was used in mild-to-moderate patients with AD to assess walking-related movements, and the authors (Della Sala, et al.) found that 40% of their series had GA.
The “Postural transition apraxia” or “apraxia of postural transitions” are other medical terms used to name GA, an uncommon subtype of Apraxia affecting the necessary movements of the trunk and lower limbs for walking, associated with problems initiating and executing these transitions. GA cases usually have problems with planning, coordinating and executing motor movements, frequently displaying hesitant, jerky movements and difficulty maintaining equilibrium, classically due to lesions on the previously cited supplementary motor regions related to ischemic stroke on both anterior cerebral artery territories, traumatic brain injury, and cognitive decline [28].
These authors concluded that the Italian translation of the AWS made a remarkable contribution to the rehabilitation of GA cases through a well-proven, culturally adapted tool for the evaluation of GA, thereby improving patient care [28,29].
Brief comments on normal pressure hydrocephalus
In 1965, Adams and collaborators documented the presence of a triad characterised by abnormal gait, Urinary Incontinence (UI) and memory impairment in Normal Pressure Hydrocephalus (NPH) with normal Cerebrospinal Fluid (CSF) pressure associated with normal pressure, dilatation of the ventricular system and improvement after ventricular shunt surgery [30].
In 1974, we reported a case presenting low-pressure hydrocephalus due to acute brain injury and Subarachnoid Haemorrhage (SAH) with GA and UI also responding to Ventriculoperitoneal Shunt (VPS), showing a normal gait and better control of the urinary sphincter. We also confirmed that the previously cited triad is not always present (60%). Later, the syndrome of Normal Pressure Hydrocephalus (NPH) described by Adams et al. was divided into idiopathic and secondary forms, with the latter caused by SAH or meningitis. In contrast, the idiopathic type was proven to be related to impairment of the CSF absorption by the arachnoid granulation at the vertex or the slurred conductance of the CSF in the subarachnoid space. On the other hand, the clinical features of idiopathic NPH (iNPH) can be explained by stretching of the periventricular corona radiata, raised intracranial pressure, ischemia or fibrosing meningitis [31], while other investigators suggested that the symmetrical enlargement of the ventricular cavities was the consequence of periventricular microvascular disease leading to hydrocephalus, rather than a dynamic effect of the CSF on the walls of the ventricular system [32].
Other authors supported that postulate based on their observation on the comorbidity of coronary syndrome, diabetes mellitus and chronic arterial hypertension [33]. It is also well known that the most successful form of treating cognitive decline is iNPH, although the rate of improvement may vary. Gait disturbance is the first clinical manifestation, along with dementia syndrome and UI [34]. Apart from the previously cited symptoms of iNPH, postural instability is another complaint of those patients and disturbance of the postural control is part of the gait disturbance [35-38].
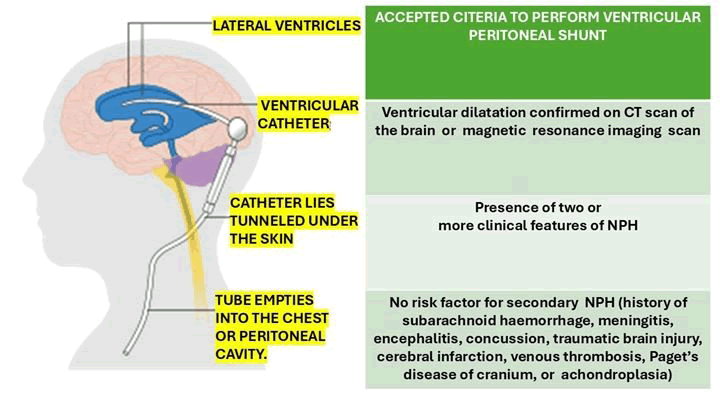
The criteria to perform a Ventriculoperitoneal Shunt (VPS) are illustrated in Figure 8.
Figure 8: The criteria to perform a Ventriculoperitoneal Shunt (VPS)
Recently, Halperin et al. studied 147 cases attending the Ospedali Riuniti of Ancona (Italy) from 2010 to 2021 who presented with iNPH, after performing confidential clinical examination and neuroimaging investigations to assess the size of the ventricles, functional status of walking, cognition, urinary continent control and mRS scores after three and six months of CSF diversion. The author concluded that no abnormal imaging was associated with cognitive improvement and that Evans indices are a key indicator, highly correlated with improvements in gait disturbance and urinary control after VPS [39].
Hayes and collaborators reported a 79-year-old lady who presented with a subacute GA, UI and cognitive decline secondary to a 2 cm vestibular schwannoma, leading to a non-obstructive dilatation of the ventricular system. This patient dramatically improved after ventriculoperitoneal shunt insertion. In this situation, hydrocephalus may be caused by decreased CSF resorption due to blockage of the arachnoid granulations in the subarachnoid space by hyperproteinorrachia [40].
Recently, other investigators from Linkoping University and the University of Gothenburg in Sweden studied the quantitative value of gait in 47 cases presenting with iNPH before and after their surgical approach, comparing it with a control group based on preoperative functional investigations. These investigators reported that step height normalised after shunt insertion. They also found that iNPH patients perambulate with diminished hip flexion, decreased velocity, shorter strides, heel strike angle and toe-off angle and had an increased time for swing and single support, compared to the healthy group [41].
In 2022, Sundström et al. conducted a systematic review of demographic factors in iNPH and found a prevalence of 400/100,000 inhabitants, while the incidence of surgical management was only 1.7/100,000 [42].
Another common feature of GA in iNPH cases is the inability to move their legs quickly during gait, whereas those patients can do so when lying in bed [43].
The American European guideline for diagnosis in 2005.
The Japanese guidelines for Mx on iNPH (2021) list three gait features; which are represented in Figure 9.
Figure 9: In the Japanese guidelines for management of iNPH (2021), three gait characteristics are highlighted: Magnetic gait and broad-based gait and the gait is characterized as unstable and slow. The gait pattern is described as duck-footed with fluctuating stride length. When turning, the steps are small and unstable and freezing of gait may be present
The classic NPH gait signs have not been widely accepted, while several studies have examined the benefits of inertial sensor systems placed on the patient’s body to quantify gait responses. The main benefits of the above-mentioned procedures are that they allow people to ambulate freely across the environment for the longest time/distance, thereby enabling the most extensive analysis of joint kinematics [44-46].
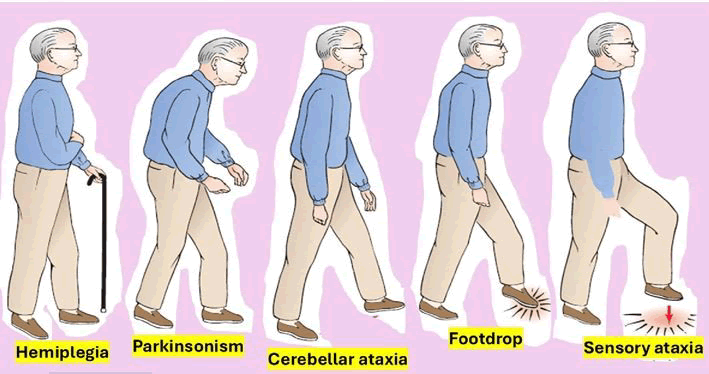
Some gait characteristics in iNPH have been differentiated from those in other conditions, as shown in Figures 1-3, documented in previous research, including impaired gait cadence and speed, increased step width and short stride length, velocity ≤ 0.83 m/s and stride length ≤ 1.02 m [47-52]. On the other hand, confirmatory tests such as the Timed Up and test (TUG) [53] and the 10-Meter Walk Test (10 MWT) [54] are also available to assess balance and other clinical features of gait. The study by Rydja et al. investigated gait features and variables related to motor response and equilibrium in iNPH patients, including quantitative evaluation before and after VPS and compared it with HI. The graphical representation of different gait abnormalities is shown in Figure 10.
Figure 10: Shows different modalities of abnormal gait
Conclusion
In conclusion based on the findings reported by Rydja et al. there is decreased flexion at the hip joint, a decreased toeâoff angle, heel strike angle, associated shorter strides, diminished speed and prolonged time for swing and single support, compared to HI. However, in these cases, after the surgical approach, a remarkable improvement in step height is observed, with it becoming almost normal in some patients. Besides that, other gait features improved, including toeâoff angle and heel strike angle, according to the TUG and the iNPH scale gait domain test.
Acknowledgment
To thanks to Prof Thozama Dubula for his unconditional support.
Ethics Statement
This review does not require ethical approval.
Patient Privacy
All patientâidentifying information has been removed to ensure anonymity.
Conflicts of Interest
Authors of this review report there is not conflicts of interest.
References
- C. Zadikoff C, A.E. Lang, Apraxia in movement disorders, Brain, 128(2005):1480-1497.
[Crossref] [Google Scholar] [PubMed]
- S.S. Della, H. Spinnler, A. Venneri, Walking difficulties in patients with Alzheimer’s disease might originate from gait apraxia, J Neurol Neurosurg Psychiatry, 75(2004):196-201.
[Google Scholar] [PubMed]
- S.N. Gowda, B. Hodis, L.K. Schneider, Apraxia, (2025).
- S. Stoll, L. Lorentz, F. Binkofski, J. Randerath, Apraxia: From neuroanatomical pathways to clinical manifestations, Curr Neurol Neurosci Rep, 25(2025):1.
[Crossref] [Google Scholar] [PubMed]
- L.L. Dutschke, K. Stegmayer, F. Ramseyer, S. Bohlhalter, T. Vanbellingen, et al. Gesture impairments in schizophrenia are linked to increased movement and prolonged motor planning and execution, Schizophr Res, 200(2018):42-49.
[Crossref] [Google Scholar] [PubMed]
- Buchmann, M. Dangel, L. Finkel, R. Jung, I. Makhkamova, et al. Limb apraxia profiles in different clinical samples, Clin Neuropsychol, 34(2020):217-242.
[Crossref] [Google Scholar] [PubMed]
- T. Vanbellingen, D. Hofmanner, S. Kubel, S. Bohlhalter, Limb kinetic apraxia is an independent predictor for quality of life in Parkinson's disease, Mov Disord Clin Pract, 5(2018):156-159.
[Crossref] [Google Scholar] [PubMed]
- Johnen, L. Brandstetter, C. Kaergel, H. Wiendl, H. Lohmann, et al. Shared neural correlates of limb apraxia in early stages of Alzheimer's dementia and behavioural variant frontotemporal dementia, Cortex, 84(2016):1-4.
[Crossref] [Google Scholar] [PubMed]
- M. Lesourd, D. Le Gall, J. Baumard, B. Croisile, C. Jarry, et al. Apraxia and Alzheimer’s disease: Review and perspectives, Neuropsychol Rev, 23(2013):234-256.
[Crossref] [Google Scholar] [PubMed]
- L.J. Buxbaum, J. Randerath, Limb apraxia and the left parietal lobe, Handb Clin Neurol, 151(2018):349-363.
[Crossref] [Google Scholar] [PubMed]
- K. Igari, M. Fujimaki, M. Mai, M. Sakuma, S. Saiki, Differences in gait apraxia due to reduced regional cerebral blood flow in the supplementary motor area in corticobasal syndrome: A report of two cases, BMC Neurol, 25(2025):304.
- Y. Balash, E.D. Mate, R. Idries, A. Eilam, A.D. Korczyn, Limb-kinetic apraxia of legs in Parkinson’s disease: Prospective clinical investigation, Clin Park Relat Disord, 12(2025):100302.
[Crossref] [Google Scholar] [PubMed]
- D. Renftle, S. Becker, K. Brockmann, T. Gasser, K. Michaelis, et al. Evaluation of the dementia apraxia test in Parkinson’s disease patients, Dement Geriatr Cogn Disord, 51(2022):271-278.
[Crossref] [Google Scholar] [PubMed]
- Yliranta, J. Nuorva, V.L. Karjalainen, R. Ahmasalo, M. Jehkonen, The dementia apraxia test can detect early-onset Alzheimer’s disease, Neuropsychology, 37(2023):44.
[Crossref] [Google Scholar] [PubMed]
- S. Jo, J.S. Oh, E.N. Cheong, H.J. Kim, S. Lee, et al. FDG-PET patterns associated with ideomotor apraxia and imitation apraxia in patients with corticobasal syndrome, Parkinsonism Relat Disord, 88(2021):96-101.
[Crossref] [Google Scholar] [PubMed]
- M.G. Maggio, M.C. Stagnitti, E. Rizzo, A. Andaloro, A. Manuli, et al. Limb apraxia in individuals with multiple sclerosis: Is there a role of semi-immersive virtual reality in treating the Cinderella of neuropsychology?, Mult Scler Relat Disord, 69(2023):104405.
[Crossref] [Google Scholar] [PubMed]
- M. Picillo, M.F. Tepedino, F. Abate, S. Ponticorvo, R. Erro, et al. Uncovering clinical and radiological asymmetry in progressive supranuclear palsy-Richardson’s syndrome, Neurol Sci, 43(2022):3677-3682.
[Crossref] [Google Scholar] [PubMed]
- I.U. Ozonder, T.D Berkol, Investigation of Apraxia in patients with schizophrenia and bipolar disorder type I, Psychiatr Danub, 35(2023):47-55.
[Crossref] [Google Scholar] [PubMed]
- Pavlidou, P.V. Viher, H. Bachofner, F. Weiss, K. Stegmayer, et al. Hand gesture performance is impaired in major depressive disorder: A matter of working memory performance?, J Affect Disord, 292(2021):81-88.
[Crossref] [Google Scholar] [PubMed]
- S. Walther, D. Alexaki, K. Stegmayer, T. Vanbellingen, S. Bohlhalter, Conceptual disorganization impairs hand gesture performance in schizophrenia, Schizophr Res, 215(2020):467-468.
[Crossref] [Google Scholar] [PubMed]
- N. Rohrbach, C. Krewer, L. Lohnert, A. Thierfelder, J. Randerath, et al. Improvement of apraxia with augmented reality: Influencing pantomime of tool use via holographic cues, Front Neurol, 12(2021):711900.
[Crossref] [Google Scholar] [PubMed]
- W. Park, J. Kim, M. Kim, Efficacy of virtual reality therapy in ideomotor apraxia rehabilitation: A case report, Medicine, 100(2021):e26657.
[Crossref] [Google Scholar] [PubMed]
- S. Walther, M. Kunz, M. Muller, C. Zurcher, I. Vladimirova, et al. Single session transcranial magnetic stimulation ameliorates hand gesture deficits in schizophrenia, Schizophr Bull, 46(2020):286-293.
[Crossref] [Google Scholar] [PubMed]
- M. Pastore-Wapp, T. Nyffeler, T. Nef, S. Bohlhalter, T. Vanbellingen, Non-invasive brain stimulation in limb praxis and apraxia: A scoping review in healthy subjects and patients with stroke, Cortex, 138(2021):152-164.
[Crossref] [Google Scholar] [PubMed]
- J.S. Meyer, D.W. Barron, Apraxia of gait: A clinico-physiological study, Brain, 83(1960):261-284.
- S.D. Sala, A. Francescani, H. Spinnler, Gait apraxia after bilateral supplementary motor area lesion, J Neurol Neurosurg Psychiatry, 72(2002):77-85.
[Crossref] [Google Scholar] [PubMed]
- C. Zadikoff, A.E. Lang, Apraxia in movement disorders, Brain, 128(2005):1480-1497.
[Crossref] [Google Scholar] [PubMed]
- M.D. Cortese, G. Cernuzio, P. Tonin, K. Priftis, F. Piccione, The assessment of walking skills: Italian version, Front Neurol, 16(2025):1579638.
[Crossref] [Google Scholar] [PubMed]
- K. Takakusaki, Functional neuroanatomy for posture and gait control, J Mov Disord, 10(2017):1.
[Crossref] [Google Scholar] [PubMed]
- R.D. Adams, C.M. Fisher, S. Hakim, R.G. Ojemann, W.H. Sweet, Symptomatic occult hydrocephalus with normal cerebrospinal-fluid pressure: A treatable syndrome, N Engl J Med, 273(1965):117-126.
[Crossref] [Google Scholar] [PubMed]
- M.J. McGirt, G. Woodworth, A.L. Coon, G. Thomas, M.A. Williams, et al. Diagnosis, treatment, and analysis of long-term outcomes in idiopathic normal-pressure hydrocephalus, Neurosurgery, 62(2008):SHC-670.
[Crossref] [Google Scholar] [PubMed]
- W.G. Jr Bradley, A.R. Whittemore, K.E. Kortman, A.S. Watanabe, M. Homyak, et al. Marked cerebrospinal fluid void: Indicator of successful shunt in patients with suspected normal-pressure hydrocephalus, Radiology, 178(1991):459-466.
[Crossref] [Google Scholar] [PubMed]
- M. Casmiro, R. d'Alessandro, F.M. Cacciatore, R. Daidone, F. Calbucci, et al. Risk factors for the syndrome of ventricular enlargement with gait apraxia (idiopathic normal pressure hydrocephalus): A case-control study. J Neurol Neurosurg Psychiatry. 52(1989):847-852.
[Crossref] [Google Scholar] [PubMed]
- S. Hakim S, R.D. Adams, The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure: Observations on cerebrospinal fluid hydrodynamics, J Neurol Sci, 2(1965):307-327.
[Crossref] [Google Scholar] [PubMed]
- Y. Nikaido, T. Akisue, Y. Kajimoto, A. Tucker, Y. Kawami, et al. Postural instability differences between idiopathic normal pressure hydrocephalus and Parkinson’s disease, Clin Neurol Neurosurg, 165(2018):103-107.
[Crossref] [Google Scholar] [PubMed]
- Y. Nikaido, H. Urakami, T. Akisue, Y. Okada, N. Katsuta, et al. Associations among falls, gait variability, and balance function in idiopathic normal pressure hydrocephalus, Clin Neurol Neurosurg, 183(2019):105385.
[Crossref] [Google Scholar] [PubMed]
- F. Lundin, T. Ledin, C. Wikkelso, G. Leijon, Postural function in idiopathic normal pressure hydrocephalus before and after shunt surgery: A controlled study using computerized dynamic posturography (EquiTest), Clin Neurol Neurosurg, 115(2013):1626-1631.
[Crossref] [Google Scholar] [PubMed]
- L. Czerwosz, E. Szczepek, J.W. Blaszczyk, B. Sokolowska, K. Dmitruk, et al. Analysis of postural sway in patients with normal pressure hydrocephalus: Effects of shunt implantation, Eur J Med Res, 14(2009):53.
[Crossref] [Google Scholar] [PubMed]
- J.J. Halperin, R. Kurlan, J.M. Schwalb, M.D. Cusimano, G. Gronseth, et al. Practice guideline: Idiopathic normal pressure hydrocephalus: Response to shunting and predictors of response: Report of the Guideline Development, Dissemination and Implementation Subcommittee of the American Academy of Neurology, Neurology, 85(2015):2063-2071.
[Crossref] [Google Scholar] [PubMed]
- Hayes, M. Wilson, A. Gontsarova, C. Carswell, Vestibular schwannoma causing normal pressure hydrocephalus, Pract Neurol, 25(2025):70-74.
[Crossref] [Google Scholar] [PubMed]
- J. Rydja, P. Pohl, A. Eleftheriou, F. Lundin, Gait characteristics in idiopathic normal pressure hydrocephalus: A controlled study using an inertial sensor system, PLoS One, 20(2025):e0317901.
[Crossref] [Google Scholar] [PubMed]
- N. Sundstrom, F. Lundin, L. Arvidsson, M. Tullberg, C. Wikkelso, The demography of idiopathic normal pressure hydrocephalus: Data on 3000 consecutive, surgically treated patients and a systematic review of the literature, J Neurosurg, 137(2022):1310-1320.
[Crossref] [Google Scholar] [PubMed]
- H. Baezner, M. Hennerici, From trepidant abasia to motor network failure-gait disorders as a consequence of Subcortical Vascular Encephalopathy (SVE): Review of historical and contemporary concepts, J Neurol Sci, 229(2005):81-88.
[Crossref] [Google Scholar] [PubMed]
- T. Backlund, J. Frankel, H. Israelsson, J. Malm, N. Sundstrom, Trunk sway in idiopathic normal pressure hydrocephalus-quantitative assessment in clinical practice, Gait Posture, 54(2017):62-70.
[Crossref] [Google Scholar] [PubMed]
- P.P. Panciani, K. Migliorati, A. Muratori, M. Gelmini, A. Padovani, et al. Computerized gait analysis with inertial sensor in the management of idiopathic normal pressure hydrocephalus, Eur J Phys Rehabil Med, 54(2018):724-729.
[Crossref] [Google Scholar] [PubMed]
- M. He, Z. Qi, Y. Shao, H. Yao, X. Zhang, et al. Quantitative evaluation of gait changes using APDM inertial sensors after the external lumbar drain in patients with idiopathic normal pressure hydrocephalus, Front Neurol, 12(2021):635044.
[Crossref] [Google Scholar] [PubMed]
- M.A. Williams, G. Thomas, B. de Lateur, H. Imteyaz, J.G. Rose, et al. Objective assessment of gait in normal-pressure hydrocephalus, Am J Phys Med Rehabil, 87(2008):39-45.
[Crossref] [Google Scholar] [PubMed]
- C. Selge, F. Schoeberl, A. Zwergal, G. Nuebling, T. Brandt, et al. Gait analysis in PSP and NPH: Dual-task conditions make the difference, Neurology, 90(2018):e1021-e1028.
[Crossref] [Google Scholar] [PubMed]
- Y.H. Lim, P.W. Ko, K.S. Park, S.K. Hwang, S.H. Kim, et al. Quantitative gait analysis and cerebrospinal fluid tap test for idiopathic normal-pressure hydrocephalus, Sci Rep, 9(2019):16255.
[Crossref] [Google Scholar] [PubMed]
- K. Mohwald, M. Wuehr, J. Decker, E.M. Asch, F. Schenkel, et al. Quantification of pathological gait parameter thresholds of idiopathic normal pressure hydrocephalus patients in clinical gait analysis, Sci Rep, 12(2022):18295.
[Crossref] [Google Scholar] [PubMed]
- T. Backlund, J Frankel, H. Israelsson, J. Malm, N. Sundstrom, Trunk sway in idiopathic normal pressure hydrocephalus-quantitative assessment in clinical practice, Gait Posture, 54(2017):62-70.
[Crossref] [Google Scholar] [PubMed]
- P.P. Panciani, K. Migliorati, A. Muratori, M. Gelmini, A. Padovani, et al. Computerized gait analysis with inertial sensor in the management of idiopathic normal pressure hydrocephalus, Eur J Phys Rehabil Med, 54(2018):724-729.
[Crossref] [Google Scholar] [PubMed]
- D. Podsiadlo, S. Richardson, The timed “Up and Go”: a test of basic functional mobility for frail elderly persons, J Am Geriatr Soc, 39(1991):142-148.
[Crossref] [Google Scholar] [PubMed]
- P. Rossier, D.T. Wade DT, Validity and reliability comparison of 4 mobility measures in patients presenting with neurologic impairment, Arch Phys Med Rehabil, 82(2001):9-13.
[Crossref] [Google Scholar] [PubMed]
Copyright: © 2026 Lourdes de Fatima Ibanez Valdes, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.