Research Article: Journal of Drug and Alcohol Research (2023) Volume 12, Issue 11
In vitro cytotoxicity of four folklore medicinal herbs of Kodanthur tribal village at Chinnar, Udumalpet, Tamil Nadu, India
Dhanabalan Rangasamy*, Lavanya Gunasekaran and Dhiviya JeyarajanDhanabalan Rangasamy, Department of Microbiology, RVS College of Arts and Science, India, Email: dhanabalan@rvsgroup.com
Received: 29-Nov-2023, Manuscript No. JDAR-23-120067; Editor assigned: 01-Dec-2023, Pre QC No. JDAR-23-120067 (PQ); Reviewed: 15-Dec-2023, QC No. JDAR-23-120067; Revised: 20-Dec-2023, Manuscript No. JDAR-23-120067 (R); Published: 27-Dec-2023, DOI: 10.4303/JDAR/236271
Abstract
Four plant species Solanum trilobatum, Spathodea campanulata, Syzygium jambos and Tylophora indica leaf extracts extracted with aqueous, methanol, ethanol and chloroform were screened for cytotoxicity in MCF-7 cells. Among 16 extracts tested significant (p<0.01) cytotoxicity was exhibited by SJCLE (74.4%), and TICLE (64.04%) with an IC50 of 95.2 μg/mL and 119 μg/mL respectively. The extract SJELE was found to exhibit 70.6% cytotoxic to MCF 7 cells but IC50 was found to higher 123.4 μg/mL compared to SJCLE and TICLE. The other extracts SCCLE, TIMLE, SJMLE, TICLE and TIELE exhibited above 60% anti-proliferative activity with their IC50 concentrations ranging from 98 μg/ mL-132 μg/mL. Furthermore, other extracts such as STELE, STMLE, and SCELE unveiled 50%-58% cytotoxicity against MCF-7 cell line at the highest concentration 300 μg/mL.
Keywords
Anticancer; Apoptosis; Cytotoxicity; Membrane blebbing
Introduction
According to estimates from the World Health Organization (WHO) in 2019,1 cancer is the first or second leading cause of death before the age of 70 years in 112 of 183 countries and ranks 3rd or 4th in a further 23 countries. Cancer’s rising prominence as a leading cause of death partly reflects marked declines in mortality rates of stroke and coronary heart disease, relative to cancer, in many countries [1]. Overall, the burden of cancer incidence and mortality is rapidly growing worldwide; this reflects both aging and growth of the population as well as changes in the prevalence and distribution of the main risk factors for cancer, several of which are associated with socioeconomic development [2]. In the development of formularies and pharmacopoeias, data on medicinal plants drive towards a new lane in the discovery of anti-cancer therapeutic agents. Herbal remedy has been approved worldwide where the plants and plant metabolites are used in the treatment of cancer. Records on plant dependent anticancer drugs from published and unpublished sources were first circulated in December 1967 by Hartwell with monumental information of 3000 species of plants with anticancer property [3]. The National Cancer Institute ratified 114,000 anticancer plant extracts from 35,000 plant species from 20 countries [4]. The isolation of the vinca alkaloids, vinblastine and vincristine from the Catharanthus roseus hosted a novel era in the use of plant constituents as anticancer agents with a paramount significance applied in the treatment of cancer at clinical level [5-7]. The discovery has paved a track to phytotherapy in cancer regimens with an advent of anticancer camptothecin derivatives, topotecan, irinotecan and etoposide considerably examined in cancer rehabilitation [8,9]. With a basic concept from the literature reviews cited above the present investigation was carried out to rule out the cytotoxic and anticancer efficacy of four medicinal plants S. trilobatum, S. campanulata, S. jambos and T. indica prevalent in the Western Ghats of Tamil Nadu.
Materials and Methods
Collection of plant material
The selected plants leaf samples were collected based on the information’s obtained from tribal communities of Kodanthur tribal village at Chinnar forest, Udumalpet and around the Western Ghats of Coimbatore district, Tamil Nadu. The voucher specimens Solanum trilobatum L. (No.1269), Spathodea campanulata P. Beauv. (No.1371), Syzygium jambos L. Alston (No.1408) and Tylophora indica (Burm.f.) Merr (No.1194) were deposited in the Department of Microbiology, RVS College of Arts and Science, Sulur for future references.
Test leaf extract preparation
Powdered and defatted fresh leaves were shade dried and further extracted with aqueous and organic solvents viz. methanol, ethanol and chloroform by cold maceration method in room temperature. Crude herb extracts were obtained after a week of soaking, filtration via. whatmann filter paper no.1, and concentration through rotary vacuum evaporator. The concentrated crude extracts were stored at 4°C for further analysis.
Cytotoxic assay
Cell line maintenance: Human breast adenocarcinoma cells (MFC-7) obtained from National Centre for Cell Sciences (NCCS), Pune was maintained in Eagles Minimum Essential Medium (EMEM) in a CO2 incubator under 37°C, 5% CO2, 95% air and 100% relative humidity. The cells were maintained by weekly passage by changing the culture medium twice a week.
Cell suspension: Single cell suspension was obtained by detaching monolayer cells with trypsin- Ethylenediaminetetraacetic Acid (EDTA). With an aid of hemocytometer, viability of cells was counted and diluted with a medium containing 5% FBS to obtain 1 × 105 cells/mL. In a 96 well microplates about 100 μL of cell suspension with plating density of 1 × 104 cells/well was plated and incubated at 37°C providing 5% CO2, 95% air and 100% relative humidity enabling for the attachment of cells. Following 24 h of incubation, the cells in the microplates were treated with different concentration of plant extracts.
Testing for cytotoxicity: About 100 μL of plant extract prepared with varying concentration ranging from minimum 300 μg/mL to 18.75 μg/mL was dispensed in to appropriate wells containing 100 μL of cell suspension followed by an additional incubation of the plates for 48 hrs. The cell suspension with DMSO and Cytoxan (CTX) (10 μg/mL) served as negative and positive controls respectively, and triplicates were maintained for all concentrations.
MTT assay: Anti-proliferative effect of test extracts on MCF-7 cells was measured using the standard colorimetric MTT (3-[4,5-dimethylthiazol-2-yl]2,5-diphenyltetrazolium bromide) assay. After 48 hours of incubation, 15 μL of MTT (5 mg/mL) in Phosphate Buffered Saline (PBS) was added to each well and incubated at 37°C for 4 h until purple precipitates were obviously visible under microscope. The medium together with MTT was then flicked, formed formazan crystals were solubilized in 100 μL of DMSO and absorbance recorded at 570 nm using a micro plate reader. The MTT assay measures the cellular mitochondrial enzymes (succinate dehydrogenases) that reduces (cleaves tetrazolium ring) the yellow water-soluble tetrazolium salt dye MTT into insoluble purple formazan. The quantity of formazan formed is directly proportional to the number of viable cells (existence of mitochondrial enzymes in the live cells) present in the plates. The percentage anti-proliferation (cytotoxicity) was determined by the following formula:
Statistical analysis
Nonlinear regression graph was plotted using drug concentration against percentage proliferation; a linear regression with trend line was used to predict the IC50 concentration of extracts that inhibits 50% proliferation of MCF-7 cells. Statistical significance was determined by one-way Analysis of Variance (ANOVA) by Dunnett’s multiple comparison tests with control (DMSO) using GraphPad Prism.
Morphological observation of MCF-7 cells
The morphological observation of cells was performed with an aid of Phase-Contrast Inverted Microscope as described [11,12]. In brief, the MCF-7 cells were plated at a density of 1 × 106 cells/mL into 6-well plates and incubated for 24 h to adhere followed by the treatment with plant extracts of determined IC50 concentrations and incubated at 37°C for 24 h. The cells were then centrifuged (300 × g for 10 min) and washed twice with Phosphate Buffered Saline (PBS) by discarding the supernatant and remaining media. About 10 μL of fluorescent dye acridine orange (AO) (10 μg/mL) was added into the cellular pellet at equal volume, kept undisturbed for 2 h and a drop of stained cell suspension was observed under fluorescent (UV) microscope within an hour. The live viable, early and late apoptotic and secondary necrotic cells were determined based on the morphology under staining. The AO is an intercalating nucleic acid-specific fluorochrome crosses the cell membrane of live and early apoptotic cells with an emission of green fluorescence, when bound to DNA. The criteria for cellular morphological identification are: Viable cell nucleus will appear green with unbroken structure, early apoptosis with bright-green condensed chromatin in nucleus, late apoptosis have dense orange areas of chromatin condensation and orange intact nucleus depicts secondary necrosis.
Results and Discussion
In vitro cytotoxicity assay
Using human mammary cancer cells (MCF-7), a total of 16 extracts from 4 plant species were evaluated for their in vitro cytotoxic activity (anti-proliferation). The assay results with significant IC50 values are depicted in Figure 1. A complete growth arrest was noticed in positive control treated with Cytoxan (Cyclophosphamide) under a minimum concentration of 18.75 μg/mL, where the drug was able to debar 92% (p<0.001) proliferation of MCF-7 cells in the culture media.
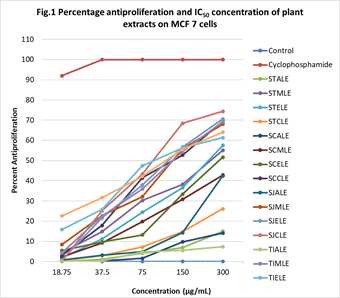
Figure 1: Graphical representation of dose dependent anti-proliferation of plant extracts
Worth noting anti-proliferation has been identified in SJCLE (74.4%), SJELE (70.6%), SCCLE (69.52%), TIMLE (69.3%), SJMLE (67.99%), TICLE (64.04%), and TIELE (61.32%) at the concentration of 300 μg/mL, where their IC50 concentrations ranged between 95 μg/mL to 132 μg/mL, though fairly high still point subtly towards selective activity. A dose dependent anti-proliferation of plant extracts was represented and the percentage of anti-proliferation were further examined by Analysis of Variation (ANOVA) with Dunnett’s multiple comparison test for the statistical significance. The extracts SJCLE and TICLE represented a highest significance (p<0.00) of antiproliferation in the MCF 7 cell line. Furthermore, the other group of extracts SCCLE, SJMLE, SJELE, TIMLE, and TIELE showed a significant (p<0.05) anti-proliferation but not comparable to Cytoxan, which could be due to the less active metabolites arresting cell proliferation. In the entire study the cell suspension with DMSO maintained as a negative control showed cell line proliferation in an uncontrollable manner. Among 4 plants the Syzygium jambos and Tylophora indica extracts excluding aqueous form were found to be more effective on cell lines inhibiting proliferation.
Cytotoxic potential of STELE and STMLE produced 57.48% and 55.11% cell death at 300 μg/mL with an IC50 concentration of 192 μg/mL and 203 μg/mL respectively. The cell proliferation restriction of STALE and STCLE indicated that much more concentration of >300 μg/mL of extract is required for significant anti-proliferative effect of MCF 7 cells.
In the entire study the highest anti-proliferative property has been observed from the extracts of S. jambos>T. indica>S. campanulata>S. trilobatum and our findings could be a comparable report with other reports previously published. Our result concurs with the antiproliferative property of S. trilobatum ethanolic leaf extracts on Ehrlich Ascites Carcinoma (EAC) cells and inhibition of mice melanoma and metastasis, which could be due to the presence of flavonoid quercetin in the plant [13-15]. The antiproliferative property of S. trilobatum was substantiated by the American cancer society which booms flavonol ‘Quercetin’ and its significance in anticancer therapy [16]. Epidemiological studies recommends dietary flavonoids and its role in reducing the risk of tumors in colon, breast, prostate, lung and pancreas [17,18]. A study submits the Saponin Fractions from S. trilobatum (SFST) has a dosedependent suppressive effect in cell proliferation [19]. A partially purified Sobatum from S. trilobatum proved cytotoxic in Dalton’s Lymphoma ascites, Ehrlich ascites and tissue culture cells (L929 and Vero) [20].
The other herb S. campanulate proved to be moderately anti-proliferative on MCF and cells. Six common human dietary sources of anticancer flavonols from methanolic leaf extracts of S. campanulata such as 1-O-caffeoyl-ẞ-D-glucopyranoside kaempferol 3-O-(2- O-ẞ-D-xylopyranosyl)-ẞ-D-galactopyranoside, kaempferol 3-O-(6-O-ẞ-L-rhamnopyranosyl)-ẞ-Dgalactopyranoside, acteoside, kaempferol 3-O-(6-O-α-Lrhamnopyranosyl)- α-D-glucopyranoside and quercetin 3-O-(2-O-ẞ-D-xylopyranosyl)-ẞ-D-galactopyranoside has been reported [21].The dietary sources of flavonols in S. campanulata might attribute to the antiproliferation in synergistic or distinctive action.
In vitro cytotoxic activity of compounds was evaluated by using SRB assay against human leukemia cancer cell lines (HL-60) [22]. The researcher isolated Stigmasta-5,22-dien- 3-ol, octadecenamide, and umbelliferone from chloroform extract that showed cytotoxicity with decreasing cell viability in a dose-dependent manner, but it was found low as compared to positive control, i.e., Adriamycin against HL-60 cell lines.
Among all extracts subjected in the study the chloroform leaf extract of S. jambos (SJCLE) revealed highest cytotoxicity against MCF-7 cells. Besides the SJELE and SJMLE unveiled cytotoxicity with IC50 of 123.4 μg/ mL and 131.4 μg/mL respectively. Previous reports on methanolic extracts of S. jambos Linn leaves were studied in vitro on Hep G2 cells, and the results demonstrated their cytotoxic effect with well-established indication of programmed cell death with the fragmentation of chromatin into units of one or more nucleosomes that form the nucleosomal DNA ladder on an agarose gel. The study’s gels unequivocally demonstrated that, at the optimal concentration, the extract-treated cells genomic DNA was significantly fragmented. The control cells were not treated and did not exhibit any fragmentation [23]. The research confirmed the extract acted on a cellular level positively affecting the apoptotic cell cycle pathway via Bcl-2 and Bax gene expression. According to, the N-heptane fraction of S. jambos reduced all cell lines viability in comparison to the negative control, which consisted of untreated cells [24]. In a previous study examined the cytotoxicity of many methanol extracts from several Syzygium species, noting that the MCF-7 cell line did not exhibit any cytotoxicity from the methanolic extract of S. jambos at 100 μg/mL [25]. Since the extract’s chemical makeup is mostly composed of phenolic chemicals, in the present in vitro study, the cytotoxicity of the chloroform, methanol, and ethanolic extracts was greater. Two acylated flavonol glycosides myricitin and myricetin in addition to quercetin were reported from the leaves of S. jambos [26,27]. Reports recommend the inhibitory property of flavonol quercetin against mice melanoma and invasive [14,16].
At 300 μg/mL the leaf extract of T. indica (TICLE) disclosed pronounceable antiproliferation on MCF-7 cells. Effective anticancer activity of T. indica acetone and ether extracts on BHK-21 cell line was reported [28]. Unique class of antitumor tylophorine analogs differing in mode of action from known antitumor drugs has been reported [29]. Polar Phenanthrene-Based Tylophorine derivatives (PBTs) N(2,3-methylenedioxy-6-methoxy-phenanthr-9- ylmethyl)-5-aminopentanol and N-(2,3-methylenedioxy- 6-methoxy-phenanthr-9-ylmethyl)-l-2-piperidinemethanol has been proved to be potentially cytotoxic against A-549 human cancer cells [30].
Herbal metabolites are always considered as a natural medication for various ailments. Natural agents from plant sources inducing apoptosis by impeding the proliferation of malignant cells can be used in cancer chemotherapy and chemoprevention. Therapeutic plants have posed a great attention in the treatment of various types of cancer considering as safer and effective anticancer agents [31]. The consumption of flavonoid rich fruits and vegetables remained to reduce the risk of cancer [32-34]. Due to additive or synergistic and rival or confounding activity of plant extracts on tumor cell lines provide an vital model for the analysis of biological mechanisms produced in clinical effects [35-37]. Similar kind of anticancer studies has aided in the identification of plant metabolites with effective anticancer activity on cancerous cells deprived of provoking effects on normal cells [38].
Morphological observation of MCF-7 cells
With the aid of microscopic technique, the original morphological criteria of apoptosis can be perceived and appraised determining the structural alterations in cells [39,40]. The microscopic examination remained the gold standard for the precise detection of apoptosis with comprehensive information about cell lines [41]. Normal inverted microscope was applied to observe the morphological changes of MCF-7 cells in the control and test plates after 72 h post treatment under 400X magnification. The present study demonstrates that the highest antiproliferative property was exhibited by S. jambos straggled by T. indica>S. campanulata and S. trilobatum via induction of membrane blebbing on MCF-7 cells inducing apoptosis. The micrographs of cells treated with higher concentration of extracts exposed distinctive morphological membrane blebbing, a process associated with apoptosis such as cell shrinkage, chromatin condensation, DNA fragmentation and apoptotic body formation (Figures 2 and 3). The apoptotic micrographs endorse the apoptosis is a consequence of chromatin margination and cytoplasm condensation with typical compaction and segregation of the nuclear chromatin.
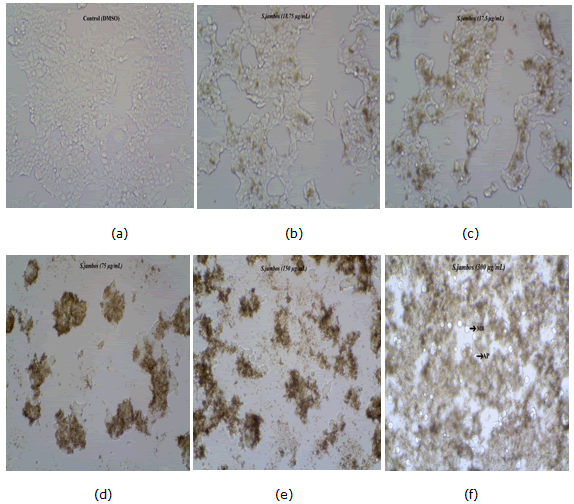
Figure 2: Micrographs of MCF 7 cells treated with chloroform leaf extracts of S.jambos examined after 48 hrs, (a) Plate 1a Control (DMSO) with confluent growth of cells; (b) Plate 1b Progression of proliferation inhibition with increase in concentration of plant extract; (c) Plate 1c Progression of proliferation inhibition with increase in concentration of plant extract; (d) Plate 1d Progression of proliferation inhibition with increase in concentration of plant extract; (e) Plate 1e Progression of proliferation inhibition with increase in concentration of plant extract; (f) Plate 1f Progression of proliferation inhibition with increase in concentration of plant extract AP: Apoptosis, MB: Membrane blebbing

Figure 3: Micrographs of MCF 7 cells treated with chloroform leaf extracts of S.jambos examined after 48 hrs, (a) Plate 2a Control (DMSO) with confluent growth of cells; (b) Plate 2b Progression of proliferation inhibition with increase in concentration of plant extract; (c) Plate 2c Progression of proliferation inhibition with increase in concentration of plant extract; (d) Plate 2d Progression of proliferation inhibition with increase in concentration of plant extract; (e) Plate 2e Progression of proliferation inhibition with increase in concentration of plant extract; (f) Plate 2f Progression of proliferation inhibition with increase in concentration of plant extract AP: Apoptosis, MB: Membrane blebbing
The morphological observation of treated cells exposed extremely impaired extra and intracellular structures by reduction in number of viable cells, in accordance with the cytotoxic property exhibited by the plant fractions; however the controls remained with confluent growth throughout the incubation period. Clear signs of apoptosis in our investigation concur with the study determining the progression of cells internal environment condensation under treatment with plant extracts [42,43]. The condensation remains accompanied by nuclear and cell outline convolutions resulting in a breach of nucleus into detached ruins to produce membrane bounded apoptotic bodies by budding of the cell. The anti-proliferative property of extracts can be evaluated by counting viable cancer cells, while the apoptogenic property shall be determined by observing typical morphological changes on apoptosis specifically membrane blebbing. Apoptosis was well evident in the micrographs of cell lines treated in a dose dependent manner of all the four plant extracts.
In the process of development and homeostasis in multicellular organisms, apoptosis remains exceedingly controlled and organized cell death process regulating variety of physiological and pathological conditions. The cytotoxicity micrographs in the present study exposed the morphological characteristics of apoptotic cells containing chromatin condensation, fragmented nuclei and DNA concurring with the previous report determining the apoptotic cellular morphological changes are due to condensation of chromatin and oligonucleosomal DNA cleavage [44]. The present study outcome indicates clearly that the selected plants S. trilobatum, S. campanulata, S. jambos and T. indica used as a folklore healthcare in traditional medicine acts via programmed cell death. Out of 4 plants selected the Syzygium jambos and Tylophora indica was found to be potent in anti-proliferative activity on MCF-7 cell lines. Additional studies are crucial to define the molecular mechanisms and its pathways to evaluate potential in vivo anticancer activity of the selected plant fractions along with active components.
Conclusion
The impact of drug efficacy in therapy always remains instigated from in vitro level before its execution to in vivo models. Plant flavonoids play a major role in disease prevention and therapy, henceforth the flavonoid rich plants are explored with great implication in the field of rehabilitation. Since the phytochemical analysis of S. trilobatum, S. campanulata, S. jambos and T. indica leaf extracts revealed the existence of flavonoids; the present cytotoxic study was executed in accordance with a concept “flavonoids as anticancer agents.” The MTT assay was instigated in the present study to accept or rule out the cytotoxic efficacy of selected plant extracts, as the method provides an accurate and reliable quantification both in cancer and noncancerous cell lines in terms of cell viability. The chloroform leaf extracts of S. jambos was found to be potent antiproliferative but not up to the level of Cytoxan. In the present study the IC50 concentration of all the active extracts with more than 60% anti-proliferation ranged from 95.2 μg/mL-132 μg/mL and the “high” IC50 values are likely may be due to very low concentration of compounds of interest, which could considerably enrich the bioactivity in the cytotoxic assay. Our study could further aim for the anticancer compound identification with an execution on in vivo models.
Funding
The infrastructure for this research was funded by DST- “Fund for Improvement of S and T Infrastructure (FIST)” Department of Biotechnology-Star College Scheme, New Delhi, Science and Technology Project Scheme under TNSCST, Chennai.
Acknowledgement
The authors, therefore, gratefully acknowledge DST-FIST, DBT Star College Scheme and TNSCST.
Conflict Of Interest
We declare that, we all authors have no conflict of interest.
References
- World Health Organization (WHO), Global health estimates 2020: Deaths by cause, age, sex, by country and by region, 2000-2019.
- O. Gersten, J.R. Wilmoth, The cancer transition in Japan since 1951, Demogr Res, 7(2002):271-306.
- J.G. Graham, M.L. Quinn, D.S. Fabricant, N.R. Farnsworth, Plants used against cancer-an extension of the work of Jonathan Hartwell, J Ethnopharmacol, 73(2000):347-377.
- M. Shoeb, S. Celik, Jaspars, Isolation, structure elucidation and bioactivity of schischkiniin, a unique indole alkaloid from the seeds of Centaurea schischkinii, Tetrahedron, 61(2005):900-06.
- M.J. Balunas, A.D. Kinghorn, Drug discovery from medicinal plants, Life Sci, 78(2005):43-41.
- J.R. Bertino, Irinotecan for colorectal cancer, Semin Oncol, 24(1997):S18-S23.
- D.J. Newman, G.M. Cragg, Natural products as sources of new drugs over the last 25 years, J Nat Prod, 70(2007):461-477.
- L. Pan, H. Chai, A.D. Kinghorn, The continuing search for antitumor agents from higher plants, Phytochem Lett, 3(2010):1-8.
- M.A. Indap, S. Radhika, L. Motiwale, K.V.K. Rao, Quercetin: Antitumor activity and pharmacological manifestations for increased therapeutic gains, Indian J Pharm Sci, 68(2006):465-469.
- T. Mosmann, Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays, J Imm Methods, 65(1983):55-63.
- J.X. Xiao, G.Q. Huang, C.P. Zhu, D.D. Ren, S.H. Zhang, Morphological study on apoptosis hela cells induced by soya saponins, Toxicol In Vitro, 21(2007):820-826.
- G. Ciapetti, D. Granchi, L. Savarino, E. Cenni, E. Magrini, et al. In vitro testing of the potential for orthopedic bone cements to cause apoptosis of osteoblast-like cells, Biomaterials, 23(2002):617-627.
- A.P. Xavier, L. Santharam, S. Panigrahi, M.S. Muthuraman, B. Pemiah, Antitumor potential of ethanolic extract of Solanum trilobatum against ehrlich's ascites carcinoma, Int J Pharm Tech Res, 5(2013):1119-1125.
- S. Caltagirone, C. Rossi, A. Poggi, F.O. Ranelletti, P.G. Natali, et al. Flavonoids apigenin and quercetin inhibit melanoma growth and metastatic potential, Int J Cancer, 87(2000):595-600.
- C.R.S. Phani, C. Vinaykumar, K.U. Rao, G. Sindhuja, Quantitative analysis of quercetin in natural sources by RP-HPLC, Inter J Res Pharm Biomed Sci, 1(2010):10-22.
- C. Sousa, B.M. Silva, P.B. Andrade, P. Valentao, A. Silva, et al. Homo-monoterpenic compounds as chemical markers for Cydonia oblonga miller, Food Chem, 100(2007):331-338.
- J.Y. Dong, L.Q. Qin, Soy isoflavones consumption and risk of breast cancer incidence or recurrence: A meta-analysis of prospective studies, Breast Cancer Res Treat, 125(2011):315-23.
- M. Messina, C. Nagata, A.H. Wu, Estimated Asian adult soy protein and isoflavone intakes, Nutr Cancer, 55(2006):1-12.
- A. Kanchana, M. Balakrishna, Anticancer effect of saponins isolated from Solanum trilobatum leaf extract and induction of apoptosis in human larynx cancer cell lines. Int J Pharm Pharm Sci, 3(2011):356-364.
- P.V. Mohanan, K.S. Devi, Effect of Sobatum on tumour development and chemically induced carcinogenesis, Cancer Lett, 112(1997):219-223.
- Y.G. Gouda, Flavonoids and phenylpropanoids from Spathodea campanulata P. beauvais leaves, Bull Pharm Sci, 32(2009):301-309.
- A. Wagh, S. Butle, D. Raut, Isolation, identification, and cytotoxicity evaluation of phytochemicals from chloroform extract of Spathodea campanulata, Futur J Pharm Sci, 7(2021):58.
- N. Thamizh Selvam, M. Acharya, V. Venkatakrishnan, S. Murugesan, Effect of methanolic extract of (Linn.) Alston leaves at intra Syzygium jambos cellular level in selective liver cancer cell line: Molecular approach for its cytotoxic activity, Adv Pharm J, 1(2016):139-43.
- L. Apaza Ticona, B. Souto Perez, V. Martin Alejano, K. Slowing, Anti-inflammatory and anti-arthritic activities of glycosylated flavonoids from Syzygium jambos in edematogenic agent-induced paw edema in mice, Rev Bras Farmacogn, 31(2021):429-441.
- G. Rocchetti, L. Lucini, S.R. Ahmed, F.R. Saber, In vitro cytotoxic activity of six Syzygium leaf extracts as related to their phenolic profiles: An untargeted UHPLC-QTOF-MS approach, Food Res Int, 126(2019):108715.
- I.I. Mahmoud, M.S.A. Marzouk, F.A. Moharram, M.R. El-Gindi, Acylated flavonol glycosides from Eugenia jambolana leaves, Phytochemistry, 58(2001):1239-44.
- A.K. Timbola, B. Szpoganicz, A. Branco, F.D. Monache, M.G. Pizzolatti, A new flavonoid from leaves of Eugenia jambolana, Fitoterapia, 73(2002):174-176.
- A. Joshi, R.S. Chauhan, Phytochemical analysis and cytotoxicity studies of Tylophora indica leaves in BHK-21 cells, Int J Trad Herb Med, 1(2013):54-58.
- C. Gopalakrishnan, D. Shankaranarayan, L. Kameswaran, S. Natarajan, Pharmacological investigations of tylophorine, the major alkaloid of Tylophora indica, Indian J Med Res, 69(1979):513-20.
- C. Gopalakrishnan, D. Shankaranarayanan, S.K. Nazimudeen, L. Kameswaran, Effect of tylophorine, a major alkaloid of Tylophora indica, on immunopathological and inflammatory reactions, Ind J Med Res, 71(1980):940-8.
- H.K. Lim, J.Y. Moon, H. Kim, M. Cho, S.K. Cho, Induction of apoptosis in U937 human leukaemia cells by the hexane fraction of an extract of immature Citrus grandis Osbeck fruits, Food Chemistry, 114(2009):1245-1250.
- S. Ramos, Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention, J Nutr Biochem, 18(2007):427-442.
- C. Kanadaswami, L. Lee, P.H. Lee, J. Hwang, F. Ke, et al. The antitumor activities of flavonoids, In vivo, 19(2005):895-909.
- W. Ren, Z. Qiao, H. Wang, L. Zhu, L. Zhang, Flavonoids: Promising anticancer agents, Med Res Rev, 23(2003):519-534.
- L.S. Adams, N.P. Seeram, M.L. Hardy, C. Carpenter, D. Heber, Analysis of the interactions of botanical extract combinations against the viability of prostate cancer cell lines, Evid Based Complement Alternat Med, (2006):117-124.
- S. Salvioli, E. Sikora, E.L. Cooper, C. Franceschi, Curcumin in cell death processes: A challenge for CAM of age-related pathologies, Evid Based Complement Alternat Med, 4(2007):181-190.
- J.Z. Luo, L. Luo, American ginseng stimulates insulin production and prevents apoptosis through regulation of uncoupling protein-2 in cultured β cells, Evid Based Complement Alternat Med, 3(2006):365-372.
- M.H. Ravindranath, T.S. Saravanan, C.C. Monteclaro, N. Presser, X. Ye, Epicatechins purified from green tea (Camellia sinensis) differentially suppresses growth of gender-dependent human cancer cell lines, Evid Based Complement Alternat Med, 3(2006):237-247.
- A.H. Wyllie, J.F. Kerr, A.R. Currie, Cell death: The significance of apoptosis, Int Rev Cytol, 68(1980):251-306.
- D.J. Taatjes, B.E. Sobel, R.C. Budd, Morphological and cytochemical determination of cell death by apoptosis, Histochem Cell BiolJan, 129(2008):33-43.
- S. Yasuhara, Y. Zhu, T. Matsui, N. Tipirneni, Y. Yasuhara, et al. Comparison of comet assay, electron microscopy, and flow cytometry for detection of apoptosis, J Histochem Cytochem, 51(2003):873-885.
- J.F.R. Kerr, A.H. Wyllie, A.R. Currie, Apoptosis: A basic biological phenomenon with wide-ranging implications in tissue kinetics, Br J Cancer, 26(1972):239-257.
- M.L. Tan, S.F. Sulaiman, N. Najimuddin, M.R. Samian, T.S. Tengku Muhammad, Methanolic extract of Pereskia bleo (Kunth) DC. (Cactaceae) induces apoptosis in breast carcinoma, T47-D cell line, J Ethnopharmacol, 96(2005):287-294.
- R.C. Sun, M.Fadia, Dahlstrom, Reversal of the glycolytic phenotype by dichloroacetate inhibits metastatic breast cancer cell growth in vitro and in vivo, Breast Cancer Res Treat, 120(2009):253-260.
Copyright: © 2023 Dhanabalan Rangasamy, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

