Research Article: Journal of Drug and Alcohol Research (2022) Volume 11, Issue 4
High-Potential of the Super-Paramagnetic of Fe3O4 with Coating by Silica-Thiol and Immobilization by Palladium on Oxidation of Benzyl Alcohols in Aerobic System
Mansour Binandeh* and Sadegh RostamniaMansour Binandeh, Department of Chemistry and Environmental Sciences, University of Maragheh, Iran, Email: mansurstrong@gmail.com
Received: 29-Mar-2022, Manuscript No. jdar-21-45421; Editor assigned: 31-Mar-2022, Pre QC No. jdar-21-45421 (PQ); Reviewed: 14-Apr-2022, QC No. jdar-21-45421; Revised: 19-Apr-2022, Manuscript No. jdar-21-45421 (R); Published: 26-Apr-2022, DOI: 10.4303/jdar/236175
Abstract
In this project, the effect of magnetic nanoparticles on the exchange and conversion of the functional group (-OH) of benzyl alcohol to the functional group (-COH) in aldehyde was investigated. For this purpose, magnetic nanoparticles (MNPs) were synthesized by chemical co-precipitation method, whose core/shell structure was confirmed by tools such as XRD, FT-IR and VSM analyzes. Next, magnetic nanoparticles (Fe3O4), for enhanced efficiency, are coated with silica and then with 3-mercapropropyl tritoxy silane (MPTES) and imidazole (to improve coating) and finally with some Pd (for immunization) over several steps in Distilled water was modified and synthesized at temperatures of 25°C-100°C. The product was named magnetic nanoparticles [Fe3O4@SiO2/SH/Pd]. The purpose of this work was to use this complex at a particle size of about 100 nm to catalyze the oxidation reaction of benzyl alcohol to benzaldehyde and its derivatives. This reaction was carried out under the following conditions: aerobic conditions with pure oxygen, non-soluble and at room temperature (25°C). Finally, the benzaldehyde products and their derivatives were synthesized one by one and analyzed by GC with TOFs optimum is 11.03 h-1. The results showed that this reaction was almost identical for all tested strains above 99%, meaning that almost all the reactants were transformed into products. This project could be suitable for the production of high quality benzaldehyde in the chemical and medical industries.
Keywords
Magnetic nanoparticles; Coating silica; Imidazole; Palladium; TetraEthyl OrthoSilicate; 3-MercaptoPropyl TriEthoxySilane
Abbrevations
(MNPs-SiO2-SH-Pd) Magnetic Nanoparticles; (Fe3O4@ SiO2) Imidazolium-Palladium, (FT-IR) Fourier Transform Infrared Spectroscopy; (XRD) X-ray Radiation Diffraction; (TEOS) TetraEthyl OrthoSilicate; (CPTES) 3-ChloroPropyl TriEthoxySilane; (MPTES) 3-MercaptoPropyl TriEthoxySilane.
Introduction
Nanotechnology is one of the most important topics in the modern scientific world. In this technology, the use of nanoparticles has many advantages that can be noted in the regular form and size of the particles [1]. Among the important nanoparticles, the use of magnetic nanoparticles is of great importance because of the unique properties of these nanoparticles by controlling the magnetic field for conducting a variety of chemical and biological reactions [2]. Proper size for nanoparticles has always been a major challenge that can be achieved with an ideal size. Therefore, efforts are being made to carefully approximate the nanoparticle size.
Magnetic properties of nanoparticles
The important thing is that magnetic nanoparticles are important because of their magnetic properties. Magnetic iron oxide nanoparticles have many applications in the fields of science, such as chemistry, physics. Iron oxide, such as magnetite (Fe3O4) and magnet (γ-Fe2O3) with biocompatibility, is widely used in many fields [3]. But it should be noted that if uncoated magnetic nanoparticles are exposed to the atmosphere, the oxide will be doubled and caused their magnetic properties to be lost under acidic conditions [4]. The project is coated with silica and thiol to prevent further oxidation of the magnetic nanoparticles.
TEOS-MPTES (3-mercaptopropyl triethoxy silane)
Magnetite, which is modified in two steps for chemical reactions, the first step in coating with silica on the magnetite surface and placing the silanol functional groups (tetraethyl orthosilate or TEOS) [5] and the second step placing the functional group 3-mercaptopropyl Source name of a group (-SH). Finally, the magnetite surface was modified with silica- mercapto. The result was a decrease in the size of the magnetic nanoparticles as well as a decrease in its magnetic energy. The magnetite-silica saturation magnetization coated with thiol group decreased to 55.05 to 20.04 (emu/ mg) [6].
The oxidation reaction of benzyl alcohol with MNPs (inion liquid)
The oxidation reaction of alcohol is not only possible with magnetic nanoparticles, but also with well-acting metals such as palladium [7]. As a catalyst, palladium is widely used in organic reactions such as Suzuki [8]. Today, the attempt to find ionic liquid as a catalyst with a suitable active surface is synthesized for various reactions and oxidation reaction of alcohols. These compounds have evolved in recent years into a research and technology center. These compounds are organic chemicals that act as a green solvent and play a very important role in reducing the use of hazardous substances that are toxic to the environment. One of the most important ionizing liquids is ionizazole containing organic molecules (imidazole ion compounds of formula C3H4N2) which is a cation. Cations and anions are used to produce a variety of ionic liquids that can be used in a wide range of ionic liquids with specific physicochemical properties. Common anions including F-, Br-, Cl- [9]. The project has two main goals: to prepare and detect Fe3O4@ SiO2/SH magnetic nanoparticles and subsequently palladium (II) stabilized on the surface of these nanoparticles, as well as perform the highest speed oxidation of benzyl alcohol and extract the best quality aldehyde products
Materials and Methods
All reagents used meet the standards and the solvent used is deionized water with a molecular weight of 18 MO/cm. Benzyl alcohol (molecular weight 108.81 g/mole, 99.9% purity), 4-methoxy benzyl alcohol (molecular weight 136.29 g/mole, 99.9 99% purity), 3-nitrobenzyl Alcohol (with a molecular weight of 152.22 g/mole, 99.9 99% purity), 4-nitrobenzylalcohol (molecular weight 154.22 g/ mole, 99.9 99% purity), 4-chloro-benzyl alcohol (with mass Molecular weight 143.69 g/mole, 99.9% purity), imidazole (C3N2H4, molar mass 68.077 g/mol), (3-choloropropyl) triethoxysilane or CPTES, (molecular weight 198.72, 97% purity), (3-mercaptopropyl) triethoxysilane or MPTES, (molecular weight 196.34 g/mol, 95% purity), hydrogen palladium (2+) tetrachloride (H2PdCl4, molecular weight 250.236 g/mol, 99% purity), of Sigma Aldrich, with reactive and non-solvent, and in aerobic conditions, by the creation of a vacuum by water by oxygen. GC analyzer (Tarbiat Modarres University of Tehran, Iran) to prove the structure of benzaldehyde and its derivatives. FeCl2.4H2O, Fe(NO3)3.9H2O, deionized water, argon gas, hydrazine (34% aqueous solution), TEOS, HCl, methanol is purchased from Sino-pharm Chemical Reagent Co. (Shanghai, China).
Synthesis of magnetic silica nanoparticles
Different mechanisms have been designed for the synthesis of hollow magnetite microspheres [10]. Chemical Co-precipitation also one of the easiest and most convenient methods of synthesis of magnetic nanoparticles with core/shell structure. The method is determined by the following formula M the same amount of iron salts (II).
To prepare Fe3O4@SiO2 nanoparticles, the Co-precipitation method was used by mixing two aqueous solutions of FeCl2 • 4H2O and (NO3)3 • 9H2O iron at a ratio of 1: 2 in sufficient amount of deionized water under argon gas flow. The mixture was stirred at room temperature for approximately 3 hours. After 3 h, the reverse micellar solution was gradually added to the mixture under severe stirring with 2 ml TEOS (tetraethyl ortho silicate). The reaction was then carried out in the laboratory for 4 hours. Finally, the prepared magnetic nanoparticles of Fe3O4@SiO2 were collected with the aid of magnetic iron and repeatedly washed with distilled water and ethanol to purify the magnetic nanoparticles and then incubated overnight in an oven at 80°C. Finally, the dried product was obtained as a yellow-brown powder.
Synthesis of magnetic nanoparticles Fe3O4@SiO2/SH/ Pd: After the synthesis of Fe3O4@SiO2 magnetic nanoparticles, the gram of this sample was weighed and dissolved in dry toluene solution. Was. And was added to the solution at a rate of 200 μL and at 24 h intervals, MPTES, and N-methyl imidazole to remove chloride. Finally, 0.006 g of palladium tetrachloride hydrogen sample was added to the mixture as a surface modifier of the magnetic nanoparticles. This took up to 6 hours. After this period, the mixture was repeatedly washed with distilled water and ethanol and placed in oven at 60°C to dry the sample. Finally, a sample of magnetic nanoparticles (Fe3O4@SiO2/SH/Pd) was obtained in the same brownish brown color (Figure 1).
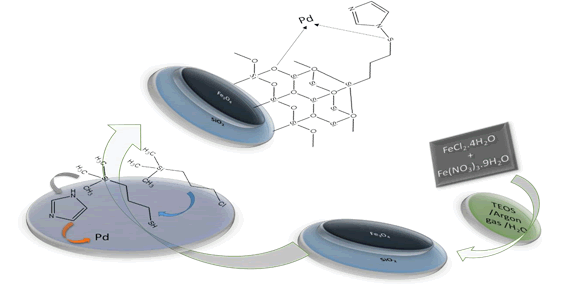
Figure 1: General route for the synthesis of Fe3O4@SiO2/SH/Pd magnetic nanoparticles.
Results and Discussion
SEM analysis of MNPs-SiO2 and MNPs-SiO2-SH-PdMagnetic nanoparticles (Fe3O4) were prepared from two oxide salts, iron (II), iron (III) with a ratio of 1: 2 and coated to prevent the surface from being oxidized to silica particles (SiO2), Also increased in medical basins. For this purpose, the size of the nanoparticles should be in the range of 1 to 100 nm, which should be less than 50 nm for biological applications. Magnetic nanoparticles were investigated and verified by a series of analyzes including TEM, SEM, XRD, FT-IR, TGA and more. This project, analyzed by XRD and FT-IR, was used to stabilize the structure of magnetite magnetic nanoparticles.
The XRD spectrum of Fe3O4@SiO2/SH/Pd
The XRD analysis, the X-ray diffraction, is an analysis that shows the number of electron transmissions using X-rays at energy levels in orbital layers and electron capacities. The synthesized Fe3O4 pixels are similar to 2 theta pixels, and in pixels between the processing of 30.1 and 35.4, 43.2, 53.7, 56.9, 62.9 and 220, 311, 422, 511, 440 cm-1 is visible in the Figure 2. Results indicate that Fe3O4 was synthesized and no changes were made in its crystalline structure. However, the pixels expressed for Fe3O4 are far from the standard pixels. Two new couriers were observed in 2 thetas of 40 and 46.7 cm-1, which are similar to the peaks of 111 and 200 cm-1, which confirm the complexation of pd to the surface of the magnetite magnetic nanoparticles with a silica substrate and factorized with the functional group (-SH). Studies courier 40 cm-1 for Pd, according to the Scherer Equation, proves the size of crystalline nanoparticles of palladium (0) at about 4 nm.
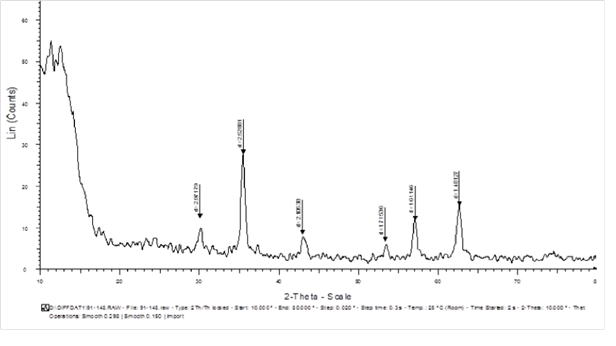
Figure 2: The XRD spectrum of Fe3O4@SiO2/SH/Pd for stabilization structure of magnetic nanoparticles.
The FT-IR spectrum of Fe3O4@SiO2/SH/Pd
FT-IR analysis, to illustrate the functional groups, as well as the covalent bond between them. For this purpose, data in the frequency range of 500 to 4000 cm-1 is used to indicate the location of the resulting functional groups. In the FT-IR spectrum, magnetite microspheres (Figure 3a) and silica-coated magnetite microspheres (Figure 3b) are characterized by a high absorbance band. 802 cm-1 is assigned to ordinary Fe3O4 bands, equivalent to the Fe-O tensile vibration modes. Subsequent coatings appeared with a silica layer, a new band of about 1,122 cm-1 (Figure 3c) that appeared to stretch the Si-O-Si band on the surface of the magnetic hollow@SiO2 microspheres. By comparing the two images Figures 3a and 3b it can be concluded that the peak of the Fe-O bond gradually became clear indicating the crystallinity of the nanoparticles. Therefore, the magnetic nanoparticle structure of the magnetite is coated with a silica layer which in addition to preventing additional oxidation and increasing the reactivity of the nanoparticles, can also give rise to a regular crystalline structure. The results for the identification of the structure of magnetic nanoparticles by the functional groups shown in Figure 4. Using Pd (II), a strong performance and modification with silica coating (Figure 3c) peaked at 1212 cm-1 and mercapto group (peak 694 and 879 cm-1) with magnetic nanoparticles as well as analysis data. The structure has become more orderly.
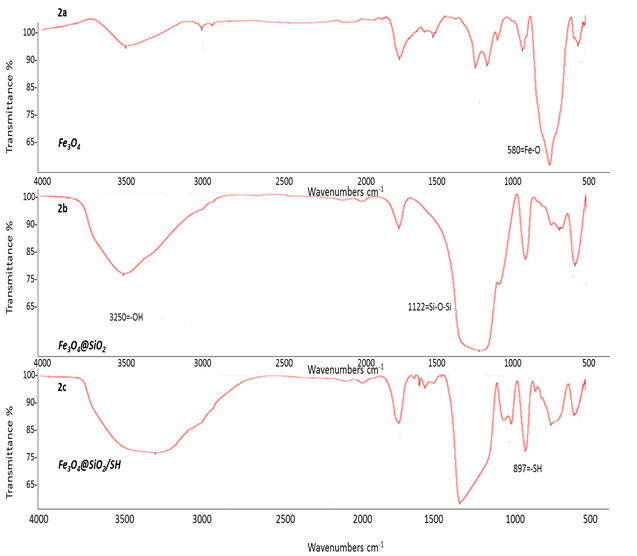
Figure 3: FT-IR analysis of magnetic nanoparticles (3a), with coated (-SiO2) (3b), and with coated (-SH) (3c), that it made increased performance level of magnetic nanoparticles.
VSM analysis
This analysis is used to measure the amount of magnetometer saturated based on the type of magnetic nanoparticles (para, dia and ferromagnetic) and the amount of magnetism in the magnetic field. Therefore, for this purpose, this analysis was used based on data suffering with criterion (emu/g) in the unit of magnetic field (kOe). Data for magnetic nanoparticles (Fe3O4) is made about 57 emu/g, 25°C -100 and PH=8-12, for silica/mercapto groups about 12, and 6 emu/g for palladium fixed on magnetic nanoparticles, which after synthesis of it (Fe3O4SiO2/SH/Pd) nanocomposite, the degree of magnetization shown is 39 emu/g. This decrease is due to the fact that magnetic nanoparticles have become superparamagnetic and their efficiency level to carry out the oxidation reaction of benzyl alcohols has increased. And also, these numbers show that the magnetic rate of magnetic nanoparticles is standard and of excellent quality, and compared to other products resulting from the project of others, about 20% more quality and adsorption properties and finally evaluated as a high-efficiency nano catalyst (Figure 4).
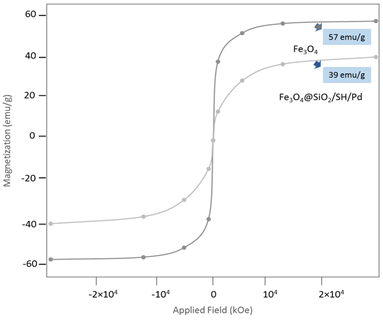
Figure 4: VSM analysis is an instrumental to measure magnetism saturated of MNPs@SiO2/SH/Pd, which it shown of 57 to 39 is decreased, with TOFs optimum 11.03h-1.
Advanced studies
Preparation of a new magnetite nanocomposite containing Pd (II) and Pd (0) in the present project and its application in the oxidation reaction of alcohols: The project uses magnetic palladium (II) magnetic nanocomposites that are fixed on its surface. By coating the silica on the surface of the magnetic nanoparticles alone it is not able to absorb organic molecules. Therefore, palladium (II) has been used to create liquid nanomaterials for the adsorption of organic molecules. Palladium has its drawbacks, if multiple reactions occur, the palladium quartz can be occupied and lose its ability to perform the next reaction. In this case the efficiency of the catalyst is reduced and in some cases it is not possible to recover and reuse the catalyst. It should therefore use a series of auxiliary coatings as ionic liquids to enhance the quality of magnetic nanocomposites for chemical and biological reactions to absorb organic molecules to their full potential at any given moment. To create such a feature, silica functional groups (SiO2) and thiol functional group (MPTES) should be used. Then, a binder such as imidazole is more applicable for the adsorption of various types of organic molecules and chemical reactions on the surface of the nanoparticles. Stable metal, such as palladium (II), can also enhance the functional level of magnetic nanoparticles by opening up its quartz spaces to allow reactions such as those under study, such as the oxidation of benzyl alcohol and its derivatives to benzaldehyde and its derivatives. To do this, one has to lose two electrons of palladium (0) in order to be palladium (II) on the nanoparticle substrate, to perform this organic reaction.
Catalytic study: This catalytic study was performed on 10 ml liters of glass reactor and the location was separated by a magnetic stirrer. The substrate and catalyst were mixed in the reactor and heated to a certain temperature. Then, oxygen was introduced into the reactor to start the system. After 24 hours of precision, the product of the desired sample of benzyl alcohols and its derivatives was analyzed separately in separate containers using gas chromatography (GC6890, using a flame ionization detector).
TOFs (turnover frequency) is calculated based on the data extracted for palladium atoms at the level of magnetic nanoparticles as follows (and optimum data is 11.03 h-1):
TOF=Converted mole of substrate (benzyl aldehydes)/ Mole of Pd atoms × Reaction time (h)
In this section, by above equation and based on the data obtained from the tour operator, by comparing the research of others [6], it can be concluded that the amount obtained from this project is for products (benzyl alcohols and its derivatives) in the range between 11-18 (ton/h) and is quite ideal.
Investigation of benzyl alcohol oxidation reaction process: In examining different methods and discussing how to react with the oxidation of benzyl alcohol and its derivatives, we conclude that the aerobic method (for the production of benzaldehydes) is economic. A schematic of the reaction is shown in (Figure 5). To investigate the oxidation of benzyl alcohol, 10 μl of benzyl alcohol and its derivatives in an aerobic condition (below the water and oxygen flow) without solvent at room temperature in the presence of 10 mg of Fe3O4@SiO2/SH/Pd magnetic nanoparticles at 25°C, 0-10 hours (all of these conditions are favorable) was evaluated. Optimization conditions include different types of catalysts and reaction time, solvents, and various types of oxidizing agents. The results obtained from the oxidation of benzyl alcohol and its derivatives for the production of benzaldehyde and its derivatives reported a high percentage (Table 1).
Table 1: Amounts of catalysts, solvents, oxidants, temperature, time and efficiency optimization.
| Compounds | Solvents | Temperature (°C) | Oxidants | Catalysis | Amount (mg) | Time (h) | Random (%) |
|---|---|---|---|---|---|---|---|
| Benzylalcohol | Dry Toulene | 80 | H2O2 | Pd | 6 | 1 | 60 |
| Benzylalcohol | Acetonitrile | 50 | MnO2 | Fe3O4@SiO2/SH | 10 | 6 | 30 |
| Benzylalcohol | Non-solvent H2O | 25 | Aerobic with O2 (Pure, 100%) | Fe3O4@SiO2/SH/Pd (Inion Liquid) | 10 | 0.25 | 99 |
| Benzylalchol Metanitro- | √ | √ | √ | √ | √ | √ | 85 |
| Benzylalcohol Paranitro- | √ | √ | √ | √ | √ | √ | 75 |
| Benzylalcohol Paracholoro- | √ | √ | √ | √ | √ | √ | 65 |
| Benzylalcohol | √ | √ | √ | √ | √ | √ | 98 |
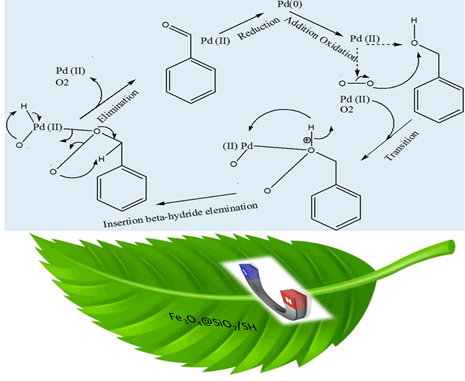
Figure 5: A representation of the benzyl alcohol oxidation reaction by magnetic nanoparticles catalysis.
GC analysis: The organic and aqueous phase analysis is performed by a GLS device using a 6890 GPS system with column size (30 m/0.205 mm/0.25 μm). As mentioned earlier, GC analysis performed with high performance to measure the percentage of benzaldehyde efficiency obtained from the oxidation reaction of benzyl alcohol obtained for 5 samples (Table 2). The in vitro results show that the yield percentages for the two samples are above 98% benzyl alcohol and para-chloro benzyl alcohol, respectively. The aerobic system is a low-cost, high-performance system in which by-products can be ignored. So aerobic exercise has been one of the best ways to do this reaction.
Table 2: GC analysis is an instrument to stabilite structure of products made of oxidation reaction benzylalcohol and its derivatives.
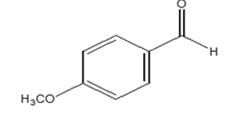
| Patterns | Products | Efficiency (%) | TOFs (TON/h-1) |
|---|---|---|---|
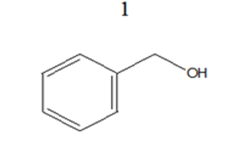 |
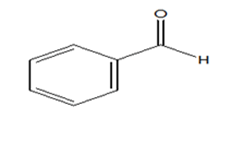 |
98 | 11.03 |
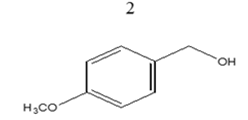 |
 |
85 | 12.27 |
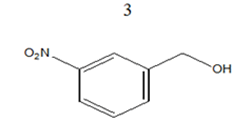 |
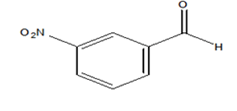 |
75 | 18.34 |
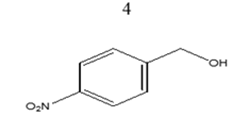 |
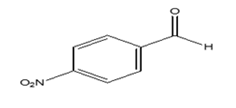 |
65 | 18.14 |
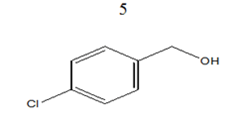 |
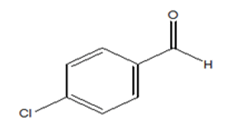 |
98 | 14.26 |
Magnetically reactive power Fe3O4@SiO2/SH/Pd magnetic nanoparticles after recycle: After investigating the optimum conditions of parameters such as catalyst, solvent, temperature and oxidizer, to convert benzyl alcohol to benzaldehyde during the oxidation reaction, we now try to investigate the performance of reusing magnetic nanoparticles several times. For this purpose, we used magnetic nanoparticles repeatedly over a period of 9 consecutive times to perform the benzyl alcohol oxidation reaction. The results showed that only 10% efficiency of the nanoparticles decreased by 9 times reuse, indicating the high performance and strength of magnetic nanoparticles as nanocatalysts (Figure 6).
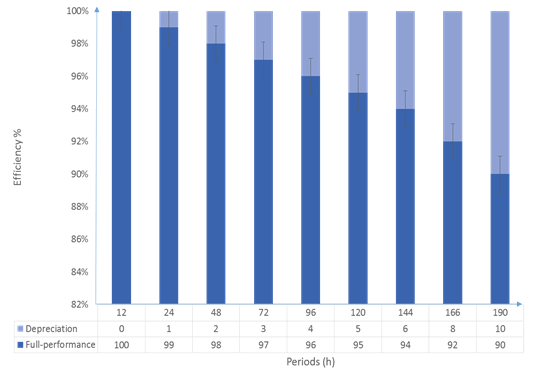
Figure 6: The stabilization and performance of again using the MNPs over time in 9 cycle.
Conclusion
In this project, after preparing the magnetic Fe3O4@SiO2 nanocomposite and stabilizing its structure, we first applied secondary thiol coating and modified it with palladium nanoparticles. Then, these nanocatalysts were tested in the oxidation reaction of benzyl alcohol. The results of these experiments were performed on 5 test samples (in 9 periods of magnetic nanoparticle reuse). The results showed that high quality aldehyde products were obtained. The high efficiency of magnetic nanoparticles is upto because, having a suitable substrate for the oxidation of alcohol, especially benzyl alcohol and, high extraction power by a magnetic field that was separated from the reaction material without destroying the products.
In addition to these two important issues, this reaction was performed under aerobic conditions (in the presence of pure oxygen), which introduced a new approach to the oxidation reaction of benzyl alcohol under anaerobic conditions by introducing a magnetic nanocomposite with the above mentioned characteristics. The ratios obtained under the optimization condition provided 10 mg of nanocatalyst under free conditions (solvent-free and 25°C) and achieved 99% efficiency over a period of 0.25 h. It is hoped that in the future these magnetic nanoparticles (Fe3O4@SiO2SH/ Pd) will be used to produce almost pure benzaldehyde and its derivatives for medical use.
Acknowledgments
This project was carried out at Maragheh University Biotechnology Lab, Iran, and I thank them.
Conflicts of Interest
Not declared.
Data Availability Statement
It’s not applicable.
Funding Statement
No funding.
References
- Z. Xu, F.S. Xiao, S. K. Purnell, O. Alexeev, S. Kawi, et al. Size-dependent catalytic activity of supported metal clusters, Nature, 372(1994), 346-348.
- M. Binandeh, Frequency of high-performance magnetic nanoparticles of mid-ampicillin, as antibacterial agents, J Antimicrob Agent, 4(2018), 162-170.
- M. Mahmoudi, S. Sant, B. Wang, S. Laurent, T. Sen, Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy, Advanced Drug Delivery Review, 63(2011), 24-26.
[Crossref] [Google Scholar] [PubMed]
- Y. Lin, H. Chen, K. Lin, B. Chen, C. Chiou, Application of magnetic nanoparticles modified with amino groups to adsorb copper ions in aqueous solution, J Environ Sci, 23(2011), 44-50.
[Crossref] [Google Scholar] [PubMed] [ResearchGate]
- D. Yang, J. Hu, S. Fu, Controlled synthesis of magnetite-silica nanocomposites via a seeded sol-gel approach, J Physical Chem, 113(2009), 7646-7651.
- Y. Zhang, Q. Xu, S. Zhang, J. Liu, J. Zhou, et al. Preparation of thiol-modified Fe3O4@SiO2 nanoparticles and their application for gold recovery from dilute solution. Separation Purification Technology, 116(2013), 391-397.
- D.I. Enache, J.K. Edwards, P. Landon, B. Solsona-Espriu, A.F. Carley, et al. Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/TiO2 catalysts, Science, 311 (2006), 362-366.
[Crossref] [Google Scholar] [PubMed]
- C. Ornelas, J. Ruiz, L. Salmon, D. Astruc, Sulphonated click dendrimer-stabilized palladium nanoparticles as highly efficient catalysts for olefin hydrogenation and Suzuki coupling reactions under ambient conditions in aqueous media, Advanced and Synthesis Catalysts, 350(2008), 48-54.
- F. Sadri, A. Ramazani, A.H. Massoudi, M. Khoobi, R. Tarasi, et al. Green oxidation of alcohols by using hydrogen peroxide in water in the presence of magnetic Fe3O4 nanoparticles as recoverable catalyst. Green Chem Let Rev, 7(2014), 257-264.
- A.H. Lu, E.L. Salabas, F. Schuth, Magnetic nanoparticles; synthesis, protection, functionalization, and application, Angew Chem Int Ed Engl, 46(2007), 1222-1228.
[Crossref] [Google Scholar] [PubMed] [ResearchGate]
Copyright: © 2022 Mansour Binandeh, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

