Research Article: Journal of Drug and Alcohol Research (2023) Volume 12, Issue 6
Hepatoprotective, Antioxidant And Anti- Inflammatory Potential Of Lentinula Edodes Extract Against Diclofenac-Induced Hepatic Damage In Wistar Rats
G. Sravan Kumar Reddy1*, B.V.V. Ravi Kumar2, M. Surya Prabha3, Uppara Venkatesh4, Ramu Samineni5 and G. Mary Ratna Anitha62Department of Pharmaceutical Analysis, Roland Institute of Pharmaceutical Sciences, Rourkela, Odisha, India
3Department of Pharmaceutical Sciences, School of Biotechnology and Pharmaceutical Sciences,Vignan’s foundation for sciences,Technology and Research,Vadlamudi,Guntur,A.P, India
4Department of Pharmacology, St. Johns College of Pharmaceutical Sciences, Yemmiganur, A.P, India
5Department of Pharmaceutics, School of Pharmacy, Sandip University, Nashik, Maharastra, India
6Department of Pharmacy, Sreedattha Institute of Pharmacy, Sheriguda, Hydetrabad, T.S, India
G. Sravan Kumar Reddy, Department of Pharmacy, Biju Patnaik University of Technology, Rourkela, Odisha, India, Email: sravan.phrma@gmail.com
Received: 03-Jul-2023, Manuscript No. JDAR-23-108971; Editor assigned: 05-Jul-2023, Pre QC No. JDAR-23-108971 (PQ); Reviewed: 19-Jul-2023, QC No. JDAR-23-108971; Revised: 24-Jul-2023, Manuscript No. JDAR-23-108971 (R); Published: 31-Jul-2023, DOI: 10.4303/JDAR/236248
Abstract
Lentinula edodes has traditionally been used to treat ulcers, bed sores, scalds and burns, hepatitis, gastritis, cancer, fungal infections, bronchial inflammation, and diabetes in Indian medicine, but its efficacy is unknown. Folklore, on the other hand, has given us numerous effective plant-based therapies. As a result, there are claims that Lentinula edodes (Omphalotaceae family) can be used to treat inflammation and hepatitis. The current study examined at the hepatoprotective, antioxidant, and anti-inflammatory properties of an alcoholic extract of Lentinula edodes. These fungi were able to inhibit the production of free radicals caused by the metabolism of the toxic substance diclofenac sodium. It also decreased diclofenac sodium-induced increases in Serum Aspartate Amino Transferase (AST), Alanine Amino Transferase (ALT), Alkaline Phosphatase (ALP), and Total Serum Bilirubin (SB), while increasing Lipid Peroxidation (LPO) and Glutathione (GSH) activities in the liver. A histopathological examination of the liver was performed to supplement the evidence from the biochemical analysis. The anti-inflammatory activity of carrageen was used to inhibit paw edoema. The results showed that the alcoholic extract of Lentinula edodes significantly (p<0.005) inhibited the carrageenan-induced paw edoema model, while the organic solvent extract (p<0.05 at 3rd h and 4th h) and the standard (p<0.01 at 3rd and p<0.001 at 4th h) when compared to the negative control. The presence of bioactive compounds such as flavonoids, tocopherol, and polyphenols reveals that Lentinula edodes has a significant protective effect against diclofenac-induced inflammation and acute hepatotoxicity
Keywords
Antioxidant; Amino transferase; Diclofenac; Hepatoprotective; Inflammation; Lentinula edodes
Introduction
Mushrooms have long been valued as highly nutritious and curative foods in many cultures around the world. People in Asian countries use mushrooms as medicine, and numerous scientific studies have been conducted on their therapeutic properties [1,2]. Mushrooms are used in Indian Ayurveda and traditional medicine. India, which is heavily reliant on agriculture, produces 620 million tonnes of agrowaste each year. India’s current agricultural situation must be transformed into a global economic power in terms of agricultural productivity by utilising new technologies. In India, any type of mushroom can be grown commercially; the most common varieties are white button, oyster, and shiitake mushrooms [3,4]. Chinese traditional medicine used mushrooms for therapeutic purposes thousands of years ago, and it still does today [5]. Physiologically active polysaccharides, amino acids, minerals, and proteins may all be found in abundance in mushrooms. Primary metabolites found in mushrooms include oxalic acid, peptides, proteins, terpenes, steroids, anthraquinones, quinolones, and derivatives of benzoic acid. The nutritionally relevant B1, B2, B12, C, D, and E tannins present in edible mushrooms make them a rich source of various nutraceuticals that display the synergistic effects of several bioactive compounds [6,7]. In recent decades, research into the medicinal properties of mushrooms has increased, leading to their promotion as natural; mini drug factories. In any event, mushrooms are a fantastic source of bioactive compounds. The list includes substances like acids, terpenoids, sesquiterpenes, polyphenols, lectins, alkaloids, lactones, glycolipids, metal chelating agents, nucleotide analogues, vitamins, glycoproteins, ergosterols, volatile organic compounds, and polysaccharides. Many different types of nutrients, including carbohydrates, proteins, vitamins, minerals, flavoring components, and bioactive components, may be found in edible mushrooms [8-10]. They are commonly used in food, flavouring agents, and traditional folk medicines. The purpose of the current study was to determine the role of alcoholic extracts of Lentinula edodes (LE) in reducing inflammation by carrageen induced inflammatory activity, antioxidant and hepatoprotection against Diclofenac- induced liver damage in wistar rats. To evaluate the hepatoprotective effect of AC, Total Serum Bilirubin (SB), Alkaline Phosphatase (ALP), Alanine Amino Transferase (ALT), Aspartate Amino Transferase (AST), and other hepatic integrity marker enzymes were measured in the serum. Additionally, the cellular levels of Lipid Peroxidation (LPO) in the form of the Malondialdehyde (MDA) protein and Glutathione (GSH) were determined [11-13]. The effectiveness of LE in preventing and treating diclofenac- induced liver histopathology in wistar rats was also demonstrated through histological reviews.
Materials and Methods
Chemicals
Dr. Reddy’s Laboratory in Hyderabad, India provided Diclofenac Sodium (DFS) 100 mg/kg. Micro Labs in Tamil Nadu, India, supplied Silymarin. Furthermore, saline was previously purchased from a nearby supplier in Hyderabad, Telangana, and India, GSN Pharmaceutical Private Limited. The following biochemical parameters were estimated using Span Diagnostics specifications kits in Surat, India: AST, ALT, ALP, and Bilirubin. Mahaveer endeavours, Hyderabad, India once supplied rat feed.
Plant material
The Telangana district of Khammam is where the fungus Lentinula edodes (LE) was found. The specimen of a plant was identified by Dr. A. Vijayabhasker Reddy of the Department of Botany at Osmania University in Hyderabad, Telangana State. They added a specimen to their herbarium. The mushrooms were thoroughly cleaned to remove any remaining soil or earthy material, then cut into thin chips and dried at room temperature in the shade before being ground to a fine powder.
Extraction
LE was collected, dried in the shade, and powdered. Each plant powder was extracted 3 times with soxhlet at room temperature (50°C-60°C) using 90% alcohol. The solvent was changed every 24 hours during the solvent extraction. The solvents from the pooled extracts were removed using a rotary evaporator under reduced pressure at 50°C-60°C to create crude extracts of Alcoholic Extract LE (AELE). Preliminary phytochemical analysis and biological activity testing were performed on the extracts.
Preliminary phytochemical studies
Khandelwal and Kokate methods will be used for preliminary phytochemical examination [14,15]. On a wide range of plant chemicals, including alkaloids, carbohydrates, fixed oils, flavonoids, glycosides, phytosterols/terpenes, proteins, saponins, and tannins/phenols, qualitative analyses are performed.
Antioxidant activity
DPPH radical scavenging activities: Ohkawa et al. investigated the DPPH radical scavenging activities of an extracts and isolated compounds [16]. In brief, 1 mL of freshly prepared DPPH (2, 2-diphenyl-1-picrylhydrazyl) solution at 0.1 mM was mixed with 0.2 mL of each of the sample solutions. After 20 minutes at room temperature, the reaction mixture was vigorously shaken, and the absorbance at 517 nm was measured. A control sample with the same volume but no test compounds or reference antioxidants was prepared, and DMSO (Dimethyl Sulfoxide) was used as a blank. The positive control in all assays was the reference antioxidant BHT (Butylatedhydroxyltoluene). The radical scavenging activity was calculated by measuring the decrease in DPPH absorbance:
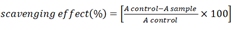
Where Acontrol is the absorbance of the control and Asample is the absorbance of the extract or fractions or standard.
Assay of FRAP: The FRAP assay was determined using the Benzie and Strain technique, with minor modifications [17-20]. It is determined by the sample’s ability to reduce the Fe (III)-TPTZ (ferric tripyridyltriazine) complex to the ferrous tripyridyltriazine Fe (II)-TPTZ at low pH. Fe (II)-TPTZ has an intense blue colour at 575 nm. 300 mM acetate buffer (pH 3.6), 10 mM TPTZ (2, 4, 6 tripyridyl S triazine) in 40 mM HCl, and 20 mM ferric chloride solution comprise the stock solutions. After mixing 25 mL of acetate buffer, 2.5 mL of TPTZ, and 2.5 mL of FeCl3.6H2O, the new working solution was ready, and the temperature was maintained at 350°C earlier than usual. In the dark, different concentrations of extracts, isolated compounds and BHT (10 g/mL-50 g/mL) were allowed to react with 2 mL of the FRAP solution for 30 minutes. At 575 nm, the absorbance was measured. The results are expressed in M Fe (II)/g and were calculated using aqueous FeSO4.7H2O 20 μM-100 μM as a calibration standard.
Inhibition of in vitro lipid peroxidation in liver homogenate: A 5% rat liver homogenate was prepared in cold TBS and 50 μl of it was used in the assay. Fresh plant tissue (0.5 g) was weighed accurately and homogenized in 1 ml of cold TBS. Aliquots of 50 μl of it were used in the assay. Ferrous sulphate at a final concentration of 10 μ moles was added to the assay medium to induce oxidation. The final volumes in the test tubes were made up to 500 μl with cold TBS. Controls were prepared for each sample, containing the respective plant extract (50 μl), liver homogenate (50 μl) and TBS to make up the final volume to 500 μl. Pro oxidant was not added to the control tubes.
A blank containing no plant extract, no liver homogenate but only FeSO4 and TBS to make a final volume of 500 μl was also prepared. An assay medium corresponding to 100% oxidant was prepared by adding all the other constituents except the plant extract and the volume was made up to 500 μl with cold TBS. The experimental medium corresponding to auto oxidation contained only the liver homogenate and TBS to make up the final volume to 500 μl. All the tubes were incubated at 37°C for 1 hour. Following the incubation period, 500 μl of 70% alcohol was added to all the tubes to stop the reaction. 1.0 ml of 10% TBA was added to all the tubes, followed by boiling in a hot water bath for 20 minutes. After cooling to room temperature, the tubes were centrifuged. To the clear supernatants collected into tubes, 500 μl of acetone was added and the TBARS was measured at 535 nm in a spectrophotometer.
Estimation of reduced glutathione: A 20% liver homogenate was obtained by homogenizing 0.5g of sample in 2.5 ml of 5% TCA. To precipitate the protein 125 μl of 25% TCA was added to 0.5 ml of tissue homogenate. The precipitated protein was centrifuged at 1000 rpm for 10 minutes. The homogenate was cooled on ice and 0.1 ml of the supernatant was taken for the estimation. The supernatant was made up to 1 ml with 0.2 M sodium phosphate buffer (pH 8.0). 2.0 ml of freshly prepared DTNB solution was added to the tubes and the intensity of the yellow colour formed was read at 412 nm in a spectrophotometer after 10 mins.
A standard curve of GSH was prepared using concentrations ranging from 2 moles-10 moles of GSH in an electronic calculator set to the linear regression mode and the values of the samples were read off it. The values are expressed as moles of GSH/g leaf.
Hepatoprotective activity
The hepatoprotective effect of the extract was evaluated using liver potential, biochemical parameters for design, Alanine Amino Transferase (ALT), Aspartate Amino Transferase (AST), Alkaline Phosphatase (ALP), Total Serum Bilirubin (SB), Lipid Peroxidation (LPO) as Malondialdehyde (MDA), and Glutathione.
Animal model and grouping: At the age of 2 months, Wistar albino rats of either sex were used in an experiment. They were divided into 6 groups of 6 animals each, and their average body weights were between 200 g and 250 g. A 24-hour light/dark cycle was used to keep the animals in standard aseptic laboratory conditions. The food is ad libitum in the form of dry pellets and water. The ethics approval committee of the institute (1684/PO/Re/S/13/CPCSEA) approved all of the animals. The study lasted 4 weeks.
Experimental setup:
• Group I: Vehicle (1 mg/kg bd.wt)
• Group II: Negative Control; (DS 100 mg/kg bd. wt, PO)
• Group III: Standard; DFS (100 mg/kg bd. wt, PO)+with simultaneous administration of Silymarin (50 mg/kg bd. wt) orally for 28 days.
wt)+with simultaneous administration of extract (100 mg/kg bd. wt) orally for 28 days.
wt)+with simultaneous administration of extract (250 mg/kg bd. wt) 28 days.
• Group VI: High dose; Receive DFS (100 mg/kg bd. wt)+with simultaneous administration of extract (500 mg/kg bd. wt) 28 days.
Histopathological study: Histopathological investigation of the liver was done according to the method described by Standish RA [20].
Anti-inflammatory activity
Carrageenan-induced paw edema test: Following 1 hour after administration of vehicle, LE extract and standard acute inflammation was produced by subplantar injection of carrageenan (0.05 mL of 1%w/v suspension), in the right hind paw of the rat. Inflammation was quantitated in terms of volume, displacement of water by edema using a digital plethysmometer 0 h before and 1 h, 2 h, 3 h, and 4 h after carrageenan injection. Diclofenac sodium 50 mg/kg was used as a standard drug. The percentage inhibition of inflammation was calculated for each group with respect to its vehicle-treated control group using the following relationship.

Where; Vo=right hind paw thickness volume (in mL) before carrageenan injection.
Vt=right hind paw thickness volume (in mL) after carrageenan injection.
Statistical analysis
Bartlett’s test was performed on each set of data to ensure that variance of the sets are homogenous [21-23]. In case of homogenous set of data ANOVA was performed to determine the treatment effects, and Dunnett’s test was employed as appropriate by using Origin Pro 7.6 statistical software. In case of heterogeneous data, it was transformed using appropriate transformation. The variance was evaluated at 5% level of significance and the values were expressed as mean ± SEM and the P<0.05 was considered as statistically significant.
Results and Discussion
Preliminary phytochemical analysis
Following the identification of the phytochemical components in the AELE extract, preliminary phytochemical testing revealed the presence of Carbohydrates, Alkaloids, flavonoids, phenols, proteins, steroids, β-carotene, and fats. The results are summarized in Table 1.
Table 1: Preparatory phytochemical screening of AELE
| Type of phytochemical constituents | Extract of LE |
|---|---|
| Carbohydrates | + |
| Alkaloids | + |
| Flavonoids | + |
| Glycosides | - |
| Tannins/phenols | + |
| Proteins | + |
| Steroids | + |
| ÃÃÂ???-carotene | + |
| Fats | + |
| -Absent, +Present | |
Antioxidant activity
DPPH radical evaluation: Due to their ability to donate hydrogen, antioxidants have the ability to change the stable radical DPPH (purple) into the non-radical form DPPH-H (yellow), acting as radical scavengers. The results of the DPPH scavenging activity for each test sample are shown in Table 2 and Figure 1. As the sample concentration (100 μg mL-1-500 μg mL-1) increased, the scavenging activity of the AELE extract and Ascorbic Acid (ASC) also increased.
Table 2: Percent inhibition of DPPH radical scavenging activity for AELE and ASC
| Sample name | Concentration | % Inhibition | Regression equation | IC50 (µg/mL) |
|---|---|---|---|---|
| AELE | 100 | 12.52 ± 1.02 | Y=0.217x+1.028 | 264.11 |
| 200 | 25.12 ± 1.12 | |||
| 300 | 46.21 ± 0.86 | |||
| 400 | 59.01 ± 0.65 | |||
| 500 | 65.18 ± 1.28 | |||
| ASC | 100 | 22.25 ± 1.10 | Y=0.2332x+7.61 | 285.52 |
| 200 | 49.82 ± 1.29 | |||
| 300 | 60.26 ± 4.30 | |||
| 400 | 72.21 ± 0.22 | |||
| 500 | 86.22 ± 1.23 |
The IC50 values were determined by AELE and ASC to be 264.11 mL-1 (Y=0.217x+1.028) and 285.22 μg mL-1 (Y=0.2332x+7.61). Based on these findings, the AELE has been identified as an effective free-radical inhibitor as well as a primary antioxidant capable of limiting free-radical damage in the body.
Figure 1: (a) DPPH radical scavenging activity of AELE and ASC; (b) FRAP scavenging activity of AELE; (c) Effect of AELE on LPO and GSH, Diclofenac induced hepatic damage in rats
Antioxidant power reduction assay (FRAP): To evaluate the antioxidant content of dietary polyphenols, the Ferric Reduction/Antioxidant Power (FRAP) assay is frequently used. Reduction properties are typically linked to the presence of compounds that break the free-radical chain by donating an atom of hydrogen. Table 3 depicts the reduction potential of fungi extracts relative to ASC, a well-known antioxidant data. The IC50 values for AELE extract and ASC were 239.67 g mL-1 (Y=0.1861x+5.39) and 181.27 g mL-1 (Y=0.2346x+7.47), respectively.
The in-vitro lipid peroxidation inhibition activity in rat liver homogenate: There has been a significant increase in MDA substance and a decrease in GSH activities in inebriated animals. Pre-treatment with silymarin (100 mg/kg b.wt) and AELE (100 mg/kg b.wt, 250 mg/kg b.wt, and 500 mg/kg b.wt) effectively kept the expansion at MDA levels and transmitted them close to the normal level, while GSH levels were increased overall (P<0.01), providing assurance against diclofenac toxicity. Table 4 shows the results.
Hepatoprotective activity of AELE
According to the assurance given by Silymarin (100 mg kg-1 b.wt; p.o.), a well-known hepatoprotective specialist, crude Alcoholic Extraction of LE (AELE) safeguarded the auxiliary uprightness of the hepatocellular film in a subordinate measurement manner as part of the study. This metabolite depletes Glutathione (GSH), causing cells to pass through (Figure 2). It is obvious that the AELE concentrate reduced all elevated levels of AST, ALT, ALP, and total serum bilirubin to normal levels, implying that plasma layer adjustment and hepatotoxin-induced hepatic tissue repair has been affected. The hepatoprotective movement of the concentrates tested is due to their similar viability, as shown in Table 5.
Table 3: Percent inhibition of antioxidant power reduction assay for AELE and ASC
| Sample name | Concentration | % Inhibition | Regression equation | IC50 (µg/mL) |
|---|---|---|---|---|
| AELE | 100 | 22.53 ± 0.62 | Y=0.1861x+5.39 | 239.67 |
| 200 | 34.24 ± 0.85 | |||
| 300 | 42.80 ± 1.46 | |||
| 400 | 49.43 ± 1.06 | |||
| 500 | 62.62 ± 0.57 | |||
| ASC | 100 | 27.68 ± 0.97 | Y=0.2346x+7.47 | 181.27 |
| 200 | 46.71 ± 1.08 | |||
| 300 | 55.01 ± 1.85 | |||
| 400 | 65.74 ± 0.83 | |||
| 500 | 76.81 ± 1.07 |
Table 4: Effect of AELE on LPO and GSH in rat liver homogenate
| Groups | LPO (nM MDA/mg protein) | GSH (µg/mg protein) | ||
|---|---|---|---|---|
| Group-I | 1.9 | 2 | 6.69 | 1.2 |
| Group-II | 5.28 | 1 | 3.25 | 1.3 |
| Group-III | 5.89 | 2.3 | 6.72 | 1.9 |
| Group-IV | 3 | 1.1 | 4.52 | 1.1 |
| Group-V | 3.41 | 1.1 | 5.28 | 1.3 |
| Group-VI | 3.68 | 1 | 5.25 | 1.3 |
Table 5: Effect of AELE on ALT, AST, ALP and SB in DS induced liver toxicity in rat
| Groups (Treated) | ALT (U/L) | AST (U/L) | ALP (U/L) | SB (mg/dl) |
|---|---|---|---|---|
| Group-I | 35.05 ± 1.01 | 52.01 ± 2.88 | 109.03 ± 6.52 | 0.54 ± 0.02 |
| Group-II | 206.62 ± 2.02a | 182.33 ± 3.02a | 215.16 ± 5.38a | 2.08 ± 0.17a |
| Group-III | 164.00 ± 1.75*** | 136.50 ± 3.68*** | 184.5 ± 3.83*** | 0.92 ± 0.07*** |
| Group-IV | 197.56 ± 5.17*** | 162.12 ± 4.06*** | 215.14 ± 5.73* | 1.65 ± 0.036*** |
| Group-V | 181.25 ± 2.75 | 140.18 ± 5.22 | 177.20 ± 2.64 | 1.22 ± 0.97 |
| Group-VI | 168.12 ± 5.23 | 130.26 ± 3.15 | 187.05 ± 5.22 | 0.91 ± 0.07 |
| Each value represents the mean ± SEM. n=6 number of animals in each group. aP<0.001 vs vehicle control, *P<0.05, **P<0.01, ***P<0.001, Compared to respective DS treated control groups. | ||||
Figure 2: Effect of AELE on ALT, AST, ALP and SB in DS induced liver toxicity in rats
Histopathological examination: The test animals were gathered after the retro-orbital plexus was punctured under mellow ether anesthesia, the animals were killed, and liver tissues were collected on the 29th day. The liver was examined histopathologically in this study in addition to the liver was examined biochemically. The effects of Alcoholic Extraction of Lentinula edodes (AELE) on ALT, AST, ALP, and SB in DS affected liver lethality in rats and microscopic analysis of Silymarin’s liver tissue. Hepatic tissue from collections subjected to doses of 100 mg kg-1, 250 mg kg-1, and 500 mg kg-1 underwent histological changes (Figure 3).
Figure 3: (a) Control animal liver tissues with normal histology; (b) Necrosis of the liver tissue of an animal treated with DS; (c) Normal hepatocytes were found in the liver tissue of DS+Silymarin-treated animals; (d) DS+100 mg/kg b.wt, p.o AELE liver tissue with normal hepatocyte arrangement; (e) DS+250 mg/kg b.wt, p.o AELE; (f) DS+500 mg/kg b.wt, p.o AELE liver tissue with normal hepatocyte arrangement
A microscopic examination of the liver tissue reveals the effects of alcohol extract of AELE on ALT, AST, ALP, and SB in DS-induced liver toxicity in rats.
Anti-inflammatory activity of AELE
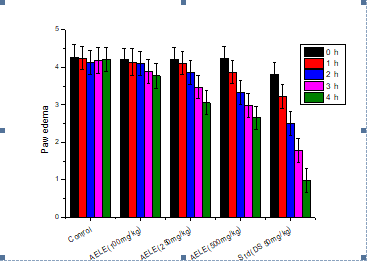
Carrageenan-induced paw edema test in Wistar rat: In carrageenan- induced paw edema model (0.1 mL of 1% freshly prepared suspension of carrageenan), the organic solvent extract showed significant inhibitory activity (p<0.05 at 3rd h and 4th h) and the standard (p<0.01 at 3rd h and p<0.001 at 4th h) when compared to the negative control (Figure 4). Maximum and minimum protections from an increase in paw volume were observed at the 4th h and 1st h respectively, for all doses and the standard diclofenac. At the peak of activity (4th h) the percentage inhibition for AELE 100 mg/ kg, 250 mg/kg, 500 mg/kg is 11.5%, 28.4%, and 37.7% respectively and % inhibition of Standard Drug (DS) at 4 h is 77.6%. The results were summarized in Table 6.
Table 6: Effect of the Alcoholic Extract of Lentinula edodes (AELE) on carrageenan-induced paw edema test in rat
| Groups | Mean paw volume ± SEM and % inhibition (I) | ||||
|---|---|---|---|---|---|
| 0h | 1h | 2h | 3h | 4h | |
| Control | 4.26 ± 0.34 | 4.24 ± 0.31 | 4.13 ± 0.32 | 4.18 ± 0.34 | 4.21 ± 0.32 |
| AELE (100 mg/kg) | 4.21 ± 0.30 | 4.14 ± 0.35 | 4.10 ± 0.31 | 3.90 ± 0.32 | 3.77 ± 0.33 |
| AELE (250 mg/kg) | 4.22 ± 0.32 | 4.11 ± 0.30 | 3.86 ± 0.31 | 3.47 ± 0.30 | 3.05 ± 0.32 |
| AELE (500 mg/kg) | 4.23 ± 0.32 | 3.87 ± 0.31 | 3.34 ± 0.32 | 2.98 ± 0.33 | 2.65 ± 0.30 |
| Standard (DS 50 mg/kg) | 3.82 ± 0.30 | 3.22 ± 0.31 | 2.51 ± 0.32 | 1.78 ± 0.33 | 0.98 ± 0.31 |
| All values are expressed as a Mean ± S.E.M, n=6. Results were analyzed using one way ANOVA followed by Dennett’s comparison multiple test. DS-Diclofenac Sodium, AELE-Alcoholic Extract of Lentinula edodes. *p<0.05 was used to indicate statistical significance when compare to control. | |||||
Figure 4: Percent inhibition of AELE concentrations and diclofenac on carrageenan-induced paw edema test in rat
Discussion
Fresh Lentinula edodes (LE) fungi were extracted three times in 90% alcohol at room temperature (50°C-60°C) on each fungi powder. The AELE extractives have a percentage yield of 16.2%. Before testing for biological activity, the extracts were subjected to preliminary phytochemical analysis. The phytochemical screening revealed that Lentinula edodes (LE, white rot fungi) extract contains a diverse array of Alkaloids, flavonoids, phenols, proteins, steroids, -carotene, and fats. They also reported the percentages of chemical composition of protein, crude fat fibres, and carbohydrates. A number of polyphenolic compounds, flavonoids, have been suggested to have antioxidant activity. When animals were pre-treated with an Alcoholic Extract of Lentinula edodes (AELE) and silymarin, the hepatic- protective properties of AELE were confirmed. This prevented the diclofenac-induced rise in transaminase and total serum bilirubin levels. The hepatoprotective activity of standard silymarin (100 mg/kg) and AELE (100 mg/kg, 250 mg/kg, 500 mg/kg) were compared. The test extract or silymarin had no effect on the increase in serum alkaline phosphatase levels. Diclofenac’s extensive liver damage reduces the rate of metabolism and other substrates for hepatic microsomal enzymes. Cytochrome P450 induction or hepatic glutathione depletion are required for DS-induced toxicity. The Alcoholic Extract of Lentinula edodes (AELE) using diclofenac reduced the elevated levels of all biochemical parameters. The significant inhibition of DS-induced liver necrosis confirmed AELE’s protective effect against experimentally induced liver damage in rats. ALT, AST, ALP, and SB are the most accurate tests for hepatic disease diagnosis. This study observed that AELE has hepatoprotective properties.
Conclusion
Historically, LE white rot fungi were used to treat ulcers, bed sores, scalds and burns, hepatitis, gastritis, cancer, fungal infections, bronchial inflammation, and diabetes. During preliminary phytochemical research, LE extracts of flavonoids, tannins, fat fibres, carbohydrates, minerals, vitamins, and phenols were discovered. AELE was discovered to have intriguing antioxidant properties such as DPPH, superoxide radical scavenging, FRAP, and hydroxy radical scavenging. Look for evidence that AELE are more active. AELE has a hepatoprotective effect in rats, as evidenced by a significant decrease in ALT, AST, ALP, SB, LPO, and an increase in GSH concentration, as well as the prevention of DS-induced histopathological changes in the liver. The maximum and minimum protections against an increase in paw volume were seen at the 4th hour with the AELE extracts and the standard diclofenac, demonstrating a moderate level of anti-inflammatory activity. In addition to previous research reports, it was clearly demonstrated that AELE had increased antioxidant, anti-inflammatory, and hepatoprotective activity when compared to ASC, Silymarin, and other plant species developing similar molecules.
Acknowledgement
The authors would like to thank the Department of Pharmacology at the Pulla Reddy Institute of Pharmacy, Hyderabad for their assistance with the research.
Conflict Of Interest
Authors have no conflict of interest to declare.
References
- M.E. Valverde, T. Hernandez-Perez, O. Paredes-Lopez, Edible mushrooms: Improving human health and promoting quality life, Int J Microbiol, 2015(2015):376387.
- P.G. Miles, S.T. Chang, Mushrooms: Cultivation, nutritional value, medicinal effect, and environmental impact, CRC Press, Boca Raton, (2004).
- T.A. Ajith, K.K. Janardhanan, Indian medicinal mushrooms as a source of antioxidant and antitumor agents, J Clin Biochem Nutr, 40(2007):157-62.
- T. Mizuno, Bioactive biomolecules and mushrooms: Food function and medicinal effects of mushroom fungi, Food Rev Int, 11(1995):7-21.
- K.H. Lee, S.L. Morris-Natschke, X. Yang, R. Huang, T. Zhou, et al. Recent progress of research on medicinal mushrooms, foods, and other herbal products used in traditional Chinese medicine, J Tradit Complement Med, 2(2012):84-95.
- A. Bhambri, M. Srivastava, V.G. Mahale, S. Mahale, S.K. Karn, Mushrooms as potential sources of active metabolites and medicines, Front Microbiol, 13(2022):837266.
- D. Agrahar-Murugkar, G. Subbulakshmi, Nutritional value of edible wild mushrooms collected from the Khasi Hills Meghalaya, Food Chem, 89(2005):599-603.
- K. Kumar, R. Mehra, R.P.F. Guine, M.J. Lima, N. Kumar, et al. Edible mushrooms: A comprehensive review on bioactive compounds with health benefits and processing aspects, Foods, 10(2021):2996.
- A. Bashir, N. Vaida, M. Ahmad Dar, Medicinal importance of mushrooms: A review, Int J Adv Res, 2(2014):1-4.
- G.S.K. Reddy, B.V.V.R. Kumar, V. Rajakumar, S. Namratha, Isolation, characteristic of bioactive compounds from Agaricus campestris and antioxidant properties, Eur Chem Bull, 12(2023): 4765-4777.
- G.S.K. Reddy, B.V.V.R. Kumar, V. Rajakumar, S. Madeesh, Antioxidant, hepatoprotective activity and anti-inflammatory of Agaricus campestris (white rot fungi) extract against diclofenac-induced hepatic damage in wistar rats, Lat Am J Pharm, 42(2023): 284-293.
- A.K. Sanapala, K.E. Kumar, Evaluation of hepatoprotective activity of Pachyrhizus erosus, extract against paracetamol induced hepatic damage in rats, Indo Am JP Sci, 3(2016):12.
- J. Redfern, M. Kinninmonth, D. Burdass, J. Verran, Using soxhlet ethanol extraction to produce and test plant material (essential oils) for their antimicrobial properties, J Microbiol Biol Educ, 15(2014):45-6.
- K.R. Khandelwal, Practical pharmacognosy techniques and experiments, Nirali Prakashan, 2003.
- C.K. Kokate, Practical pharmacognosy, Vallabh prakashan, (2005):107-111.
- C.S. Alisi, O.A. Ojiako, C.G. Osuagwu, G.O.C. Onyeze, Free radical scavenging and in-vitro antioxidant effects of ethanol extract of the medicinal herb Chromolaena odorata linn, Br J Pharm Res, 1(2011):141.
- I.F.F. Benzie, J.J. Strain, The Ferric Reducing Ability of Plasma (FRAP) as a measure of "antioxidant power": The FRAP assay, Anal Biochem, 239(1996):70-6.
- S. Selvi, P. Umadevi, S. Suja, K. Sridhar, P. Chinnaswamy, Inhibition of in vitro Lipid Peroxidation (LPO) evoked by Calocybe indica (milky mushroom), Anc Sci Life, 26(2006):42-5.
- T.E. Tipple, L.K. Rogers, Methods for the determination of plasma or tissue glutathione levels, Methods Mol Biol, 889(2012):315-24.
- R.A. Standish, E. Cholongitas, A. Dhillon, A.K. Burroughs, A.P. Dhillon, An appraisal of the histopathological assessment of liver fibrosis, Gut, 55(2006): 569-78.
- R. Devi, S. Namratha, S.A. Kumar, K.E. Kumar, Antiinflammatory activity of aqueous leaf extract of Blumea mollis, IJAP, 2(2011):487-491.
- M.T. Mansouri, A.A. Hemmati, B. Naghizadeh, S.A. Mard, A. Rezaie, et al. A study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats, Indian J Pharmacol, 47(2015):292-8.
- C. Lalanne, M. Mesbah, Measures of association, comparison of means or proportions, biostatistics and computer-based analysis of health data using SAS, Elsevier, (2017):39-75.
[Crossref]
Copyright: ©2023 G. Sravan Kumar Reddy, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.