Research Article: Journal of Drug and Alcohol Research (2026) Volume 15, Issue 2
Area Postrema Syndrome and Novel Approaching of Drug Therapy
Lourdes de Fatima Ibanez Valdes1 and Humberto Foyaca Sibat2*2Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa
Humberto Foyaca Sibat, Department of Neurology, Nelson Mandela Academic Hospital, Walter Sisulu University, South Africa, Email: humbertofoyacasibat@gmail.com
Received: 02-Jan-2026, Manuscript No. JDAR-26-171190; Editor assigned: 06-Jan-2026, Pre QC No. JDAR-26-171190 (PQ); Reviewed: 20-Jan-2026, QC No. JDAR-26-171190; Revised: 18-Feb-2026, Manuscript No. JDAR-26-171190 (R); Published: 25-Feb-2026, DOI: 10.4303/JDAR/236498
Abstract
Objectives: The main aim of this study is to discuss the last information released into the medical literature regarding to drug therapy in patients with Area Postrema Syndrome (APS) reported in the available medical literature.
Methods: A wide searching of the medical literature of the following database PubMed/MEDLINE, Scopus, and Embase databases performed to find articles related to novel information on APS and its diagnostic and therapeutic procedures. From 01st, January 1989 to 30th, January of 2026, we searched the medical literature, following the PRISMA guidelines. The authors searched the scientific databases, Scopus, Embassy, Medline, and PubMed Central using the following searches: “area postrema” OR “area postrema syndrome” OR “refractory nausea and vomiting” OR “intractable nausea, hiccups and vomiting”, OR “treatment/management of APS”.
Results: Searching literature retrieved 149 articles. 101 duplicated publications identified and after reviewing the titles and abstracts removed, forty‐eight publications selected. After applying the inclusion/exclusion criteria forty articles excluded; therefore, eight studies investigated the role of therapeutic procedures in the assessment of APS selected.
Conclusions: Only eight studies reported novel therapeutic procedures to control refractory nausea and vomiting due to APS that we discussed in this article. Based on bibliography investigation done, the best therapeutic drugs to control refractory nausea and vomiting due to APS are: Granisetron, Aprepitant, Netupitant, Palonosetron and Olanzapine prescribing alone or in combination according to the clinical respond and under safe supervision.
Keywords
Area postrema; Area postrema syndrome; Drug therapy; Refractory nausea and Vomiting
Introduction
The Area Postrema (AP) was first described by Magnus Gustaf Retzius in 1896. He described it as an anatomical structure found at the dorsal region of the brainstem, in the caudal part of the fourth ventricle. The AP is a circumventricular organ located at the caudal area of the floor of the fourth ventricle overlying the inferior region of vagal trigone while facing the foramen of Magendie and rostral to the obex, which plays a crucial role in regulating nausea and vomiting, supported by its unique Blood-Brain Barrier (BBB) permeability and extremely large connectivity [1].
More specifically, AP is a paired protuberance located at the cauda/dorsal aspect of the fourth ventricle in the medulla oblongata, which contains permeable Capillary Vessels (CV) and sensory neurons capable of identifying circulating chemical elements travelling through the blood flow and transducing them into the neural network. Because of the anatomical position of AP next to the nuclei of the tractus solitarius bilaterally, it functions as a sensory transducer, modulating blood-brain autonomic functions, including the identification of neurohormones involved in the pathophysiology of thirst, hunger, and vomiting, and the control of arterial blood pressure. This structure has links to a medical condition known as Area Postrema Syndrome (APS), characterised by unexplained episodes of intractable nausea, vomiting and hiccups [2].
Although lesioned AP has the preventive capacity of vomiting in response to most emetic medications, it cannot play a remarkable role in cases presenting vomiting caused by motion or afferent inputs from the vagal nerve or radiation-induced vomiting. Proper expression of the AP triggers nausea and vomiting by projecting electrical input to the adjacent nucleus tractus solitarius. The AP acts as a critical hub in the brainstem regulation of nausea and vomiting, integrating signals from the gastrointestinal tract, serum, and multiple CNS circuits.
The clinical manifestations of poorly controlled nausea and vomiting imply a large differential diagnosis. Therefore, finding the central or peripheral origin of that symptom is essential to obtain a good history of present complaint and a physical examination, in addition to medical information about medications, substance use, toxic exposures, and medical conditions [3].
On the other side, the total volume of the Blood Flow (BF) at the AP is remarkably large compared with the surrounding areas. In contrast, the circulation velocity of its blood components is quite slow, leading to increased capacity to modulate the passage of almost all circulating elements across the membrane, such as neurotransmitters and neurohormones [4].
In 2024, Bruce et al. proved that the dorsal vagal complex in the brainstem works as a modulatory centre for energy and glucose homeostasis by monitoring hormonal and nutrient changes [5].
In 2023, Zhang et al. documented that metformin increases kidney GDF15 synthesis, elevating plasma GDF15 levels, and induces GFRAL expression in the AP to diminish feeding and weight in rats [6].
Area Postrema Syndrome (APS) is clinically characterised by intractable nausea, singultus and vomiting, and it can be associated with other pathological infectious, inflammatory, demyelinating, and autoimmune conditions, among others. Negative AQP4, MOG, and GFAP antibody testing with autoimmune encephalitis can also be associated with APS [7].
Last year, Munoz-Zuniga and collaborators reported the first patient presenting APS associated with systemic lupus erythematosus [8].
Recently, other authors reported a 63-year-old woman presenting with an APS secondary medullary ischemic stroke near the AP leading to intractable nausea, vomiting and hiccups without focal neurological signs [9].
Courtney et al. reported a 76-year-old man presenting APS characterised by intractable hiccoughs, nausea, vomiting, with several watershed cerebellar strokes and a medullary ischemic stroke related to Giant Cell Arteritis (GCA), who responded exceptionally well to corticosteroids, weekly subcutaneous tocilizumab, an IL-6 inhibitor, resolving both his APS and GCA [10].
Recently, Li and Teng documented that APS is a relatively uncommon symptom of Autoimmune Glial Fibrillary Acidic Protein Astrocytopathy (A-GFAP-A) based on their findings on 7 of 75 (4 females and 3 males (9.3%)) GFAP-IgG positive cases with a median age of onset of 42 years presenting the APS [11].
Materials and Methods
A wide searching of the medical literature of the following database PubMed/Medline, Scopus, and Embase databases performed to find articles related to novel information on APS and its therapeutic procedures.
From 01st, January 1989 to 30th, January of 2026, we searched the medical literature, following the PRISMA guidelines (2020 statement). The authors searched the scientific databases, Scopus, Embassy, Medline, and PubMed Central using the following searches: “area postrema” OR “area postrema syndrome” OR “refractory nausea and vomiting” OR “intractable nausea, hiccups and vomiting”, OR “treatment/management of APS”.
Search strategy
A systematic search done through the following databases: PubMedCentral/Medline, Scopus, and Embase to identify publications related to novel information related to APS and treatment.
Only articles published in English, Spanish and Portuguese selected. Editorials, preclinical studies, and conference proceedings excluded.
Selection of study
Both investigators (LdeFIV and HFS) separately reviewed the abstracts and titles of the selected publications and independently reviewed the full-text versions of the identified articles to determine their eligibility for inclusion. Medical investigations and therapeutic approaches delivering information on APS studies selected for inclusion in this systematic study. Furthermore, publications without a clear protocol of confirmatory diagnosis, papers with lack of analysis, publications with incomplete data or without mention exact number of patients were excluded.
Selection criteria
The following criteria were included: Articles with detailed pathogenesis and/or drug management. Exclusion criteria were applied:
• Inaccessibility to full text.
• Articles with unclear pathogenesis.
• Lack of relevant clinicopathological data.
• Non-original studies (i.e., editorials, letters, conference proceedings, book chapters).
• Animal model studies;
• Non-/Spanish/Portuguese/English studies’ .
• Studies without results on therapeutic management.
The papers that were not thoroughly assessed were removed.
Data extraction and quality assessment
The studies’ quality assessment including applicability concerns and risk of bias was categorized as good, poor, fair, or reasonable, in agreement with the National Institutes of Health criteria and the Quality Assessment of Diagnostic Accuracy Studies version 2 (QUADAS-2) evaluation. Quality evaluation made separately by both authors, and disagreements resolved by scientific discussion and final agreement.
Data collection, extraction, and bias assessment
All abstracts and titles with the inclusion criteria were revised by two before cited authors to collect relevant information for the review. For each publication selected in the review, data concerning to author’s name, age, year of publication, country of information’ source, type of the study, drug therapy implemented. The selected data from eligible publications were introduced into an updated Excel software program.
Outcome measures
Our plan aimed to select the most relevant publications relating to the clinical benefit and real-world effectiveness of different diagnostic/therapeutic procedures for APS.
Statistical analysis
Statistical investigation was done through XLSTAT (add-on for Microsoft Excel, version 2021.4.1, Addinsoft SARL) and RStudio.
Results and Discussion
Literature search
Searching literature retrieved 149 articles. 101 duplicated publications identified and after reviewing the titles and abstracts removed, forty eight publications selected. After applying the inclusion/exclusion criteria forty articles excluded; therefore, eight studies investigated the role of therapeutic procedures in the assessment of APS selected.
To provide an accurate management of this searching, the authors used a QUADAS-2 evaluation to determine low/ moderate risk of bias for all publications.
Brief comments on Area Postrema Syndrome (APS)
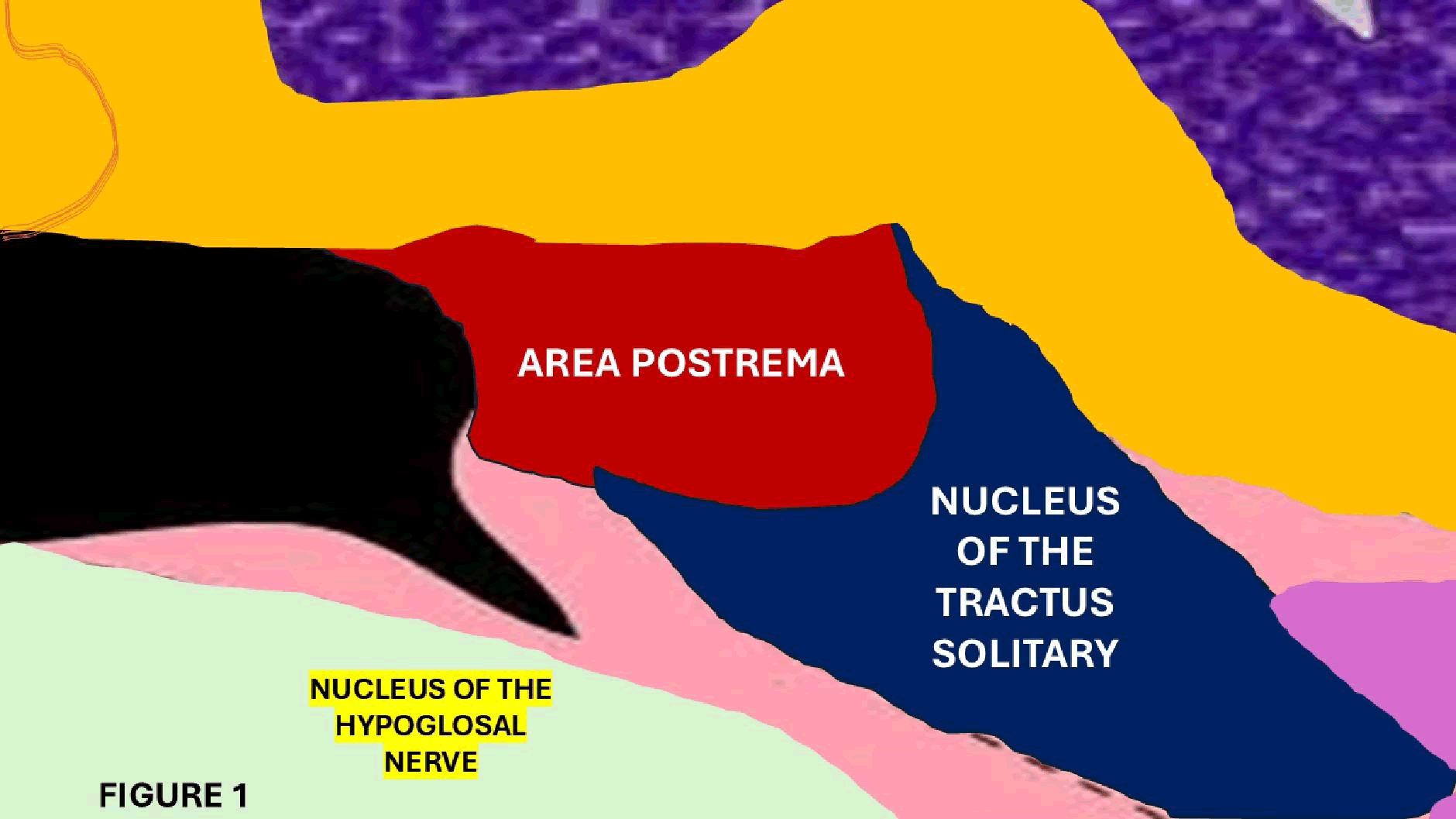
There are highly specialized ependymal cells in the AP, which are completely different from other ependymal cells located along the ventricular walls and central canal in the spinal cord. The surrounding anatomical structures around to AP represented in Figure 1.
Figure 1: Graphical representation of the AP and surrounding tissues
Anatomically, the AP separated by the funiculus separans (FS-a thin, semi-transparent ridge) from the vagal trigone, which overlies the dorsal nucleus of the vagal nerve located on the upper part of the floor of the IV ventricle. However, the FS and AP share a thick ependyma-containing tanycyte covering, which participates in chemoreception and in the transport of neurochemicals across the CSF in both directions from surrounding elements such as neurons, glial cells, and blood vessels.
Because of the proximity of the AP to the ventricles, it is a circumventricular organ located just before the obex in the lower part of the medulla oblongata at the caudal aspect of the floor of the fourth ventricle. There are crucial differences between the Capillary Vessels (CV) at the AP. At the same time, those located in the ventral subregions are impermeable to CV elements.
In contrast, those in the dorsal/medial regions of the AP are permeable to CV elements. They are called: Fenestrated sinusoidal capillaries forming a portal system as a vascular component, while the cellular component is made by flattened ependymal cells (tanycyte) made up of thick ependyma), glial cells, and small neurons, which make this particular area remarkably important in the autonomic control of many pathophysiological mechanisms, including controlling feeding and metabolism and the cardiovascular system. A part of the dorsal vagal complex, the AP is the vomiting centre. The AP, together with the NTS and the DMNX, where afferent vagal nerve fibres arrive.
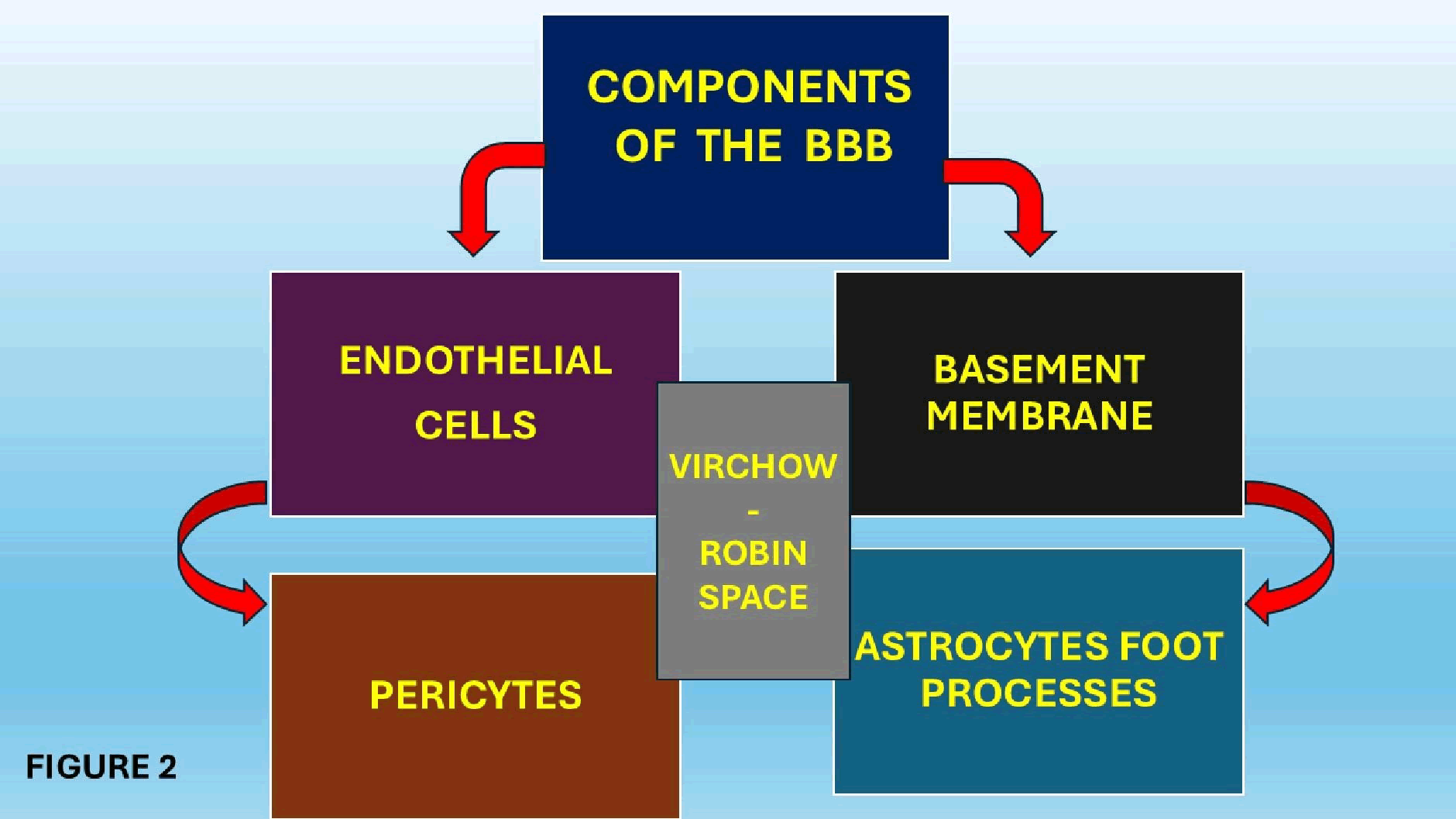
The AP characterized by its lack of blood-brain barrier which main components are illustrates in Figure 2.
Figure 2: Schematic representation of the BBB
Where the BBB is missing, it compensated for by a tanycyte barrier. On top of that, the density of the subregional CV is highest close to the ventricular surface, and twice near the Dorsal Nucleus of the Vagus nerve (DNV) and the Nucleus of the Tractus Solitarii (NTS) [12]
The AP connects to the main autonomic control centres and the NTS in the brainstem after stimulated by afferent autonomic visceral impulses from the humoral centres, the gastrointestinal tract, and other peripheral trigger zones. The same authors have documented that AP is one of the components of the dorsal vagal complex, together with NTS and DNV, which are crucial termination sites for afferent fibres from the tenth cranial nerve.
A feeling of sickness with a tendency to vomit with nausea is mainly induced via stimulation of the AP through its connection to the NTS due to a response to several afferent emetic inputs usually preceding the pathways of vomiting, including detection of blood toxins, chemoreceptor trigger zone in response to emetic drugs, but excepting the activating mechanism caused by motion.
Surgical approach/damage/injury to the AP is also known as central vagotomy due to its role in the brain’s capacity to monitor the body’s physiological status via the afferent vagal nerve. On the other side, AP is also involved in the mechanism for the detection of lithium chloride intoxication [13].
Fortunately, lesional damage of the AP can confirmed by neuroimaging and its common clinical features characterized by refractory nausea, singultus, and vomiting [14].
Other concomitant clinical manifestations caused by neighbouring anatomical lesions in the brainstem and/or cerebellum.
APS does not respond appropriately to first-line antiemetics. Nevertheless, in most cases, it must manage by confirming and managing its underlying aetiology.
In APS, the commonest aetiology of brainstem-localized hyperemesis is its comorbidity with Neuromyelitis Optica Spectrum Disorder (NMOSD), which is a well-known autoimmune demyelinating condition in response to autoantibodies to Aquaporin 4 (AQP4), which is a protein activated on astrocyte end feet processes and ependymal cells surrounding CVOs, including the AP. Therefore, we suggested that improving the clinical issues of APS caused by NMOSD must include adequate management of the autoimmune process and its inflammatory response, including glucocorticoids, intravenous immunoglobulins, monoclonal antibodies, and plasmapheresis [15].
The investigation of choice to confirm lesions in the AP is a brain MRI. Recently, Lara-Garcia et al assessed 3D FSE Cube T2WI, 3D FSE Cube FLAIR, and 3D DIR MRI sequences to improve AP confirmation in thirty-five patients with and without demyelinating disease, such as Multiple Sclerosis (MS), and thirty-five patients with other non-demyelinating CNS diseases (ND-CNSD). They foundthat the AP not identified in 27.6% of the 3D FSE cube T2WI sequences. At the same time, it confirmed in 99% of the 3D FSE cube FLAIR sequences and 100% of the 3D DIR sequences, concluding that the 3D DIR sequence showed superior performance in identifying the AP [16]. Brief comments on nausea and vomiting in area postrema syndrome.
Since the 19th century, few investigators have been documenting the presence of a “vomiting centre” at the floor of the IV ventricle [17,18]. On the other hand, since the second half of the last century, it has reported that the location of the “chemoreceptor trigger zone” for nausea is around the AP in the medulla oblongata [19].
We also hypothesized that the histological features of the AP, including its fenestrated capillary vessels and the lack of a proper BBB, facilitate the transmission of humoral signals from the blood vessels directly to the selected neurons located at the AP, including the nucleus of the tractus solitarii and the dorsal lateral nucleus of the vagal nerve.
Nausea is a warning signal of an unpleasant visceral sensation, often associated with poisoning or motion, leading to reflexive actions such as vomiting (or emesis) in response to noxious stimuli Table 1 [20].
| Receptor | Distribution | Mechanism | Treatment implications |
| Adenylate Cyclase Activating Polypeptide Receptor (ADCYAP-R) | Central: AP/NTS, vestibular nucleus. | ADCYAP-1R is a G-protein coupled receptor that responds to PACAP, a neuropeptide involved in the stress response and trigemino-vascular system. Activation of the ADCYAP-Rs in the AP causes activation of signaling pathways in the AP/NTS involved in the emetic response. PACAP is released in response to LPS, with the downstream effect of modulating aversive feeding behaviours. |
Antagonism could be a future potential target. There are very early in vitro and pre-clinical studies using small- molecule antagonists. |
| Aquaporin-4 (AQP-R) | Central: Astrocytes and ependymal cells, particularly at the end feet processes surrounding CVOs-there is a very high density within the AP. Also found in the NTS and hypothalamus. | Gate water transport in the CNS and are critical to the integrity of the BBB. Regulate the fine osmotic balance within the CNS. In NMOSD, severe oedema results from antibodies directed at AQP-4, leading to intractable nausea and vomiting if the lesion is near the AP. |
Could be a potential future target for selectively limiting brain oedema following CNS injury and thus limit some effects from the disruption in BBB integrity. |
| Calcitonin Gene-Related Peptide (CGRP-R) |
Peripheral: Enteric nervous system, vagal afferents, trigeminal nerve, dorsal root ganglia. Central: Parabrachial nucleus (PBN), NTS, AP. |
CGRP is released by trigeminal and vagal afferents, playing a key role in the nausea induced by migraines. CGRP-containing neurons in PBN are activated by noxious stimuli and project to the NTS via glutamatergic synapses. Circulating gastric hormones can cause the release of CGRP in the AP, which stimulates the PBN. The PBN sends projections to subcortical areas that process emetic and interoceptive signals. This leads to food aversion behaviours in animal studies. | CRGP antagonists are commonly used for migraine treatment and have- a well-established role in central pain sensitization. The prominent central action may indicate a potentially useful adjuvant for central causes of N/V. |
| Cannabinoid-1 (CB1-R) | Central: AP, NTS, DMX, hypothalamus, hippocampus. Peripheral: Enteric nervous system, GI epithelial cells. | CB1 is a retrograde neurotransmitter system that acts to limit serotonin release centrally, thus attenuating nausea and vomiting through suppression specifically within the NTS. Administration of THC directly into the brainstem mitigates CINV in ferrets through activation of CB-Rs. Some, albeit weak, evidence to suggest downregulation of CB1 leads to cannabis hyperemesis syndrome. Alternative hypotheses include desensitization with repeated exposure. In general, receptor activation stimulates feeding behaviour through interactions between the NTS and PBN. | Used in CINV, especially in refractory situations. Although there are complex central interactions, CB1-Rs are a potential target for antiemetic effects. |
| Dopamine-2 (D2-R) | Central: Distributed widely in the AP, NTS, DMX. Peripheral: Vagal afferents, GI tract. Also located throughout the cortex, and may play a role in signaling from the DVC to higher-order structures, such as the insula. |
Receptors in the AP are activated by circulating emetic stimuli, leading to intracellular signaling cascades and activation of the vomiting reflex. Receptors in the DMX are involved in modulating gastric tone and phasic contractions. |
Demonstrated efficacy in certain central causes of nausea and vomiting, such as migraines. Current anti- dopaminergic agents act centrally (contributing to extrapyramidal side-effects) but also exert some 5HT-3 blocking effect. |
| GDNF Family Receptor- Alpha-Like (GFRAL) | Central: AP, NTS, PBN. | Activated by GDF-15, a stress response cytokine, the production of which is stimulated by metformin, systemic inflammation, various tumours, and chemotherapy. Stimulation reduces feeding behaviour and can induce nausea and vomiting. GDF-15 has wide- ranging effects throughout the body, but activation of GFRAL is specific to AP/NTS. | GDF-15 monoclonal antibody-abated cisplatin induced nausea and vomiting in non-human primate model. Also thought to play a role during pregnancy-related nausea and vomiting. GFRAL antibody has been used in mouse model to attenuate tumor-associated cachexia. |
| Ghrelin-R | Central: AP, NTS, hypothalamus. Peripheral: Vagus nerve, GI mucosal cells. | Stimulates receptors directly within the AP to activate feeding behaviours in animal models. Also modulates AP/NTS activity through stimulation by vagal afferents. Requires a functional AP to mediate effects, as seen in lesional studies. Peripherally it has prokinetic effects. | Agonists are currently being investigated for both prokinetic and anti- nausea uses. |
| GLP-R | Central: Mainly in the AP and NTS, but can also be found in PBN. Peripheral: Vagal afferents. | GLP-1 is released by gut enterochromaffin cells after meals and exerts some effect locally to slow GI transit. However, it also mediates effects through glutamatergic projections in the AP/NTS, leading to appetite suppression. Weight-loss effect of agonists is mediated through the AP/NTS, as central lesions in animal models ablate the effects of these peptides. Activation mediates avoidance of aversive substances in mouse models, likely through action in the PBN. | Action of gastric peptides can be avoiding use of these agonists with- respect to any central cause of nausea and vomiting is reasonable. Antagonism may be a potential target for future therapeutics. |
| Glucose-Dependent Insulinotropic Polypeptide (GIP-R) | Central: AP, NTS. Peripheral: GI vagal afferents. | Like many gastric peptides, GIP readily crosses the BBB to exert effects at the AP. Agonism causes ergic neurons in the AP/NTS to inhibit emetic pathways in animal models of CINV. Further studies show GIP agonism does not diminish the weight- loss effects of anorectic peptides such as GLP-1 agonists. |
Additional example of a centrally acting gastric hormone that attenuates nausea, with some pre-clinical utility in CINV models. When administered with the anorectic peptides being used for weight loss, reduces nausea and vomiting without affecting hypophagia. |
| Histamine Receptor type 1 (H1) | Central: AP, NTS, DMV, vestibular nuclei. Peripheral: Vagal afferents. | H1-R mediated depolarization of neurons within the AP/NTS increases glutamatergic signaling, which leads to vestibular-induced nausea and vomiting. Glutamatergic signaling within the NTS is highly involved in vestibulo- autonomic reflex stimulation. Activation of vagal afferents in the GI tract relays signals that induce the emetic response via the AP/NTS. In animal studies, intraventricular administration of histamine caused vomiting that was prevented with either AP ablation or anti-histamine pretreatment. | Many antiemetic agents have broad central H1 and M1 receptors in the vestibular circuit (i.e., prochlorperazine, diphenhydramine). |
| Interleukin-6 (IL-R) | Central: AP, hypothalamus,- organum vasculosum of the lamina terminalis. | Activation of IL-6 receptors, particularly in the AP, stimulates excitatory signaling within the NTS to activate the emetic pathway. Stimulation in pre- clinical models induces cachexia. In mouse tumor models, IL-6R- blocking antibodies ameliorated anorexic effects and prolonged lifespan. Partial mechanistic explanation for how systemic inflammation can mediate nausea, vomiting, and centrally mediated food avoidance. | This is one of the potential pathways by which steroids can reduce central nausea, as they limit the release of cytokines onto CVOs. |
| Mu Opioid Receptor | Central: AP, NTS.- Peripheral: Vagal afferents, GI tract. | Stimulation of the mu opioid receptor causes vomiting in animal models that is reduced with AP ablation but not with vagotomy. Three mechanisms: (1) Direct stimulation of DVC. (2) Slowing of GI transit. (3) Stimulation of the vestibular apparatus. | Demonstrates the importance of avoiding opioids in central nausea and Advances in Intracellular Emetic Signali vomiting. |
| Muscarinic Acetylcholine Receptor Type- 1 (M1) | Central: AP, NTS, vestibular nuclei. Peripheral: Vagal afferents in the GI tract. | Cholinergic signaling induced by vestibular stimuli leads to excitatory signaling within the AP/NTS. In feline studies, intraventricular administration of M1 agonist caused emesis, and this was prevented with ablation of AP. |
Scopolamine is commonly used in motion-sickness and post-operative nausea, acting centrally via suppression of vestibular signaling. |
| Neurokinin-1 (NK-R) | Central: AP, NTS, DMX. Peripheral: myenteric plexus in the enteric nervous system, dorsal root ganglia, sympathetic ganglia. | Substance P is released from enterochromaffin cells in the gut, although to a far- lesser extent than- 5HT-3. Substance P release activates NK-1R’s in the AP/NTS, stimulating emetogenic reflex. Predominantly central action in CINV, as evidenced by ferret studies showing no response to NK-1R antagonists when the ability to cross the BBB is removed. |
Widely accepted as effective for treatment of the delayed phase of CINV. Also relevant in gastroparesis, with no impact on GI motility, indicating prominent centrally acting effects. Also thought to be involved in the complex and poorly understood cyclic vomiting syndrome. NK-1R antagonists given during the prodromal phase significantly reduce the duration of vomiting episodes. |
| Neuropeptide-2 (NPY-R) | Central: AP and NTS. Peripheral: Vagal afferents. | Peptide YY (PYY) is secreted from GI neuroendocrine cells. NPY2-R, stimulated by three ligands: PYY, NPY, and pancreatic polyprotein process. PYY crosses the BBB, and then exerts effects on receptors in the AP, predominantly to evoke anorectic effects in animal models. These effects cease with AP ablation. Also involved in the cardiovagal response, leading to vasoconstriction and hypotension. | Further evidence of- the strong connection between gut hormones and the AP. Potential future target. |
| Oxytocin-R | Central: AP, NTS, insula. Peripheral: Vagal afferents in the GI tract. | Stimulation has anorectic effects through increasing gastric pressure via vagal preganglionic signaling in the DVC and vagal afferents. There is evidence that activation can alter behaviour through insular signaling pathways (food intake, etc.). |
There are very early pre-clinical studies looking at oxytocin-R antagonists for modulating nausea and visceral pain sensations. |
| Prostaglandin E2-R | Mainly found peripherally in the GI tract and vagal afferents. Central: AP, NTS. | Prostaglandin E2 (PGE2) is derived from cell membrane phospholipids in response to inflammation, especially in the GI tract. Circulating PGE2 stimulates receptors in the AP and leads to activation of nausea and vomiting pathways. PGE2 levels correlate with nausea and vomiting during pregnancy. | One of the many mechanisms by which glucocorticoids exert antiemetic effects: by reducing formation of prostaglandins during inflammation. |
| Serotonin Type- 3 (5HT-3-R) | Peripheral: High receptor concentration on vagal afferents. Central: Receptors also located centrally, to a lesser extent, in the NTS, AP, and DMX. | 5HT-3 is mainly released from enterochromaffin cells in the GI tract. Initial cascade relays visceral sensation to the area postrema (AP) and NTS through vagal afferents which modulate glutamatergic signaling within the NTS. 5HT-R is an ion channel that causes depolarization and the release of multiple neurotransmitters such as Substance P and cholecystokinin onto the NTS. | 5HT-3 antagonists are one of the mainstays of treatment of CINV and nausea and vomiting in general. Dexamethasone has been shown to modulate vagal 5HT-3 activity. |
| Serotonin Type 4 (5-HT4-R) | Peripheral: Mainly within the GI tract (intestinal epithelial cells and enteric neurons). Central: AP, NTS, DMX. | Activation of the receptor enhances acetylcholine release and stimulates GI motility, overall prokinetic effects. Central receptors modulate the sensory input from the vagus- nerve, reducing nausea. | Many medications available have either full or partial agonist effects. Useful in gastroparesis, irritable bowel syndrome, and postoperative ileus. |
| Toll-like Receptor 4 (TLR-4) | Central: Found in CVOs, notably the AP. | Activated by inflammatory mediators such as LPS, increasing permeability of CVOs and neuroinflammation. Thought to play a role- in central parasitic infections (trypanosomiasis). | Additional mechanism by which steroids act to reduce nausea and vomiting in the setting of inflammation. |
| Transient Receptor Potential Vanilloid-1 (TRPV-1) | Central: AP, NTS Peripheral: Presynaptic myelinated and unmyelinated vagal afferents, dorsal root ganglia, trigeminal ganglia. | Activated by capsaicin, heat, and low pH. Activation leads to release of substance P onto NK-1 R’s, both peripherally and in the AP/NTS. Also causes the release of GCRP. Overstimulation leads to depletion of substance P and both peripheral and central internalization of the NK-1 Rs, providing an antiemetic effect. Central TRPV-1 receptors are also thought to be partially stimulated by cannabinoids, which may explain the antiemetic effects of cannabis. |
Capsaicin cream is used in cannabis hyperemesis syndrome. The mechanism is somewhat unclear, although it likely involves peripheral desensitization of depletion of substance P. Potential adjuvant to modulate central NK-1 R activity through peripheral depletion of substance P. |
Table 1: Receptors expressed in peripheral and central neural circuits implicated in mediating the sensation of nausea and execution of the vomiting response
The symptom of upset sensation can also block the sense of appetite, as evidenced by a long-lasting rejection of triggering stimuli such as certain odours, flavours, and tastes related to the experience of nausea. However, under specific circumstances induced by anaesthetic agents, nausea may serve as a protective mechanism which can be extremely undesirable and lead to complications [21]. In contrast, nausea associated with vascular headaches, gastrointestinal conditions and side effects of chemotherapy does not exhibit its protective role, and it can lead to a pathological disorder of its own [22].
We hypothesize that, in cases complaining of nausea related to Area Postrema Syndrome (APS), the most common anti- nausea drugs, such as those targeting substance P receptors, histamine receptors, and serotonin receptors, do not have the capacity to block the neuronal discharge of the AP’s efferent pathways.
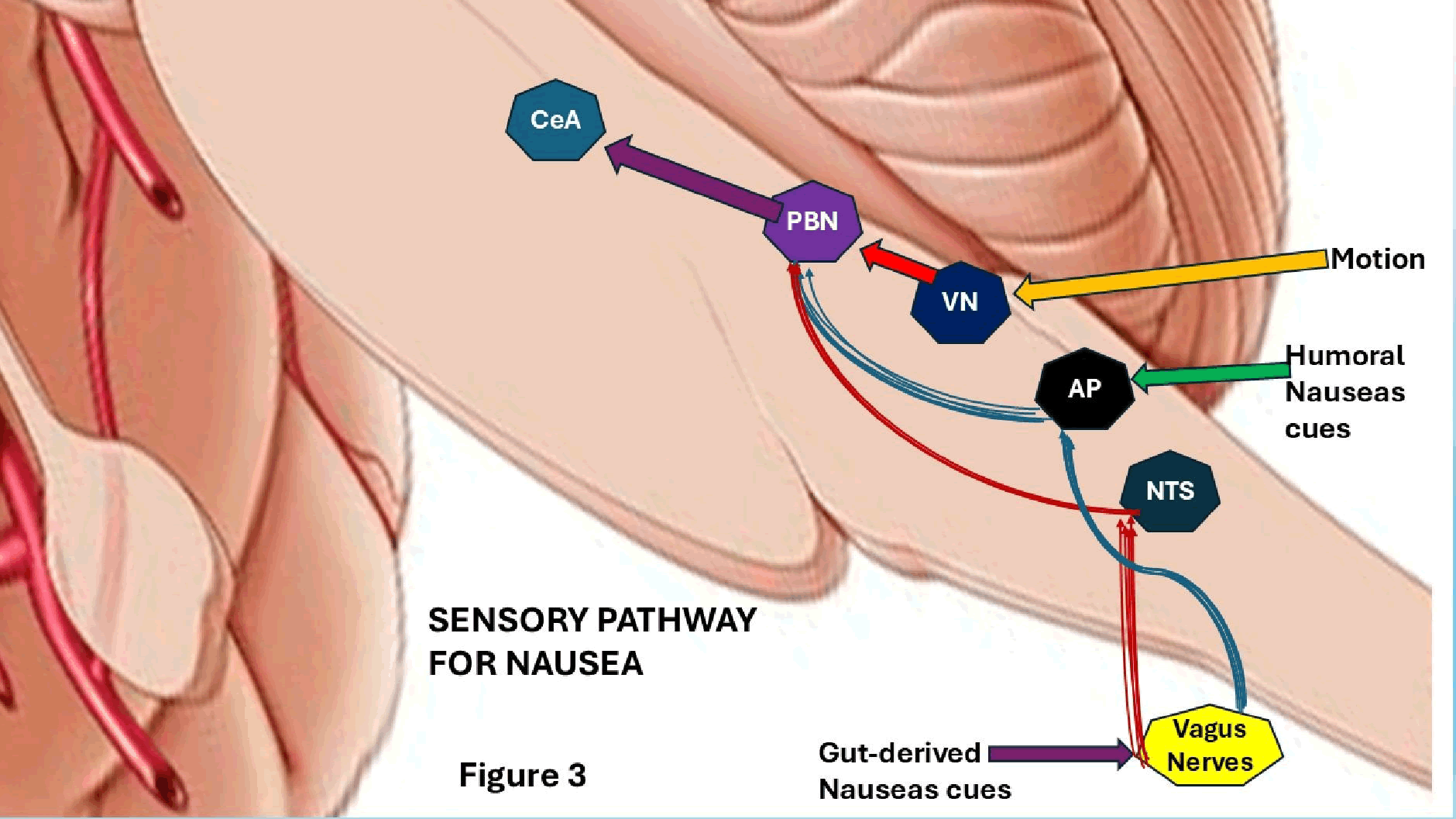
As is well known, exteroceptive stimuli from the olfactory and somatosensory pathways (vestibular/motion sickness and visceral/poisoning) may induce nausea – an interoceptive sensation – via those peripheral pathways, which can activate CNS centres, as represented in Figure 3.
Figure 3: Visceral pathways have the vagus nerve, responsible for detecting nausea signals from the gut, and the area postrema, which directly senses humoral cues and may receive secondary inputs from the vagus nerve. The vestibular pathway encompasses the Vestibular Nucleus (VN) that mediates motion sickness. It seems that all sensory pathways converge at the Parabrachial Nucleus (PBN) and Central Amygdala (CeA), which are crucial for aversive learning
Recurrent syncope is an associated clinical manifestation of APS related to Neuromyelitis Optica Spectrum Disorders (NMOSD) confirmed by MRI of the brain [23].
The AP is a sensory circumventricular organ with direct access to blood flow and CSF through its highly permeable fenestrae CV, thereby providing rapid humoral access to metabolites and peptides [24].
The AP is the chemoreceptor trigger region for nausea as a sensitive area for nausea-related hematogenous stimulus to induce vomiting [25]
Emetic stimuli, such as apomorphine, act on AP to provoke vomiting, whereas other stimuli, such as veratrum alkaloid toxins, do not [26].
Therefore, we hypothesized that there are distinct types of specialized excitatory (calcitonin receptor)/inhibitory neurons in the AP, with distinct thresholds for the molecular components of stimuli, and that these neurons respond differently to adjacent vagal nuclei. Other authors have documented it based on their findings about two groups of neurons showing differentiated behavioural effects and anatomical projections, such as 1. GLP-1 Receptor (GLP1R) is composed of two smaller excitatory clusters and 2 smaller (SLC6A2+ or GFRAL+) or large (GLP1R+) agonist neuronal populations, which express solute carrier family 6 member 2 (SLC6A2) [27].
Other authors have documented that GLP1R-expressing and non-aversive neuronal populations (CALCR+) signal to the midbrain hub and the Parabrachial Nucleus (PBN), conveying most interoceptive and somatosensory information [28-30].
On the other hand, PBN neurons densely innervated by GLP1R AP, which express Calcitonin Gene-Related Peptide (CGRP) [31].
Nevertheless, PBN CGRP+ cells create monosynaptic connections in the Central Amygdala (CeA), leading to malaise-induced conditioned avoidance of novel flavours and lack of appetite [32,33]. The CeA activated by novel flavours, which stabilized and reactivated by malaise signals via the PBN CGRP+ neuronal inputs, leading to learned avoidance of the novel flavours despite the delayed malaise responses [34].
Because, the before cited lack of BBB in the AP, it can directly identify humoral information through some specialized and activated superficial cellular receptors to modulate feeding, gut neurohormones such as amylin, ghrelin, GLP-1, Glucose-dependent Insulinotropic Polypeptide (GIP) and peptide YY, angiotensin, GDF15 (playing a role in food allergy-induced aversion), NF-kB pathway and COX2 expression [35-40].
Brief comments on the vagal neural pathway
The vagal nerve plays a remarkable role in in the interconnections between the brain and the gut through the gut-brain axis, there are important functional and genetic variations among vagal sensory neurons and other cranial nerves which support its somatic-visceral functions including a variety of sensory neurons able to provide innervation of the intramuscular array endings, intra- ganglionic laminar endings, and mucosal endings in the gut [41,42].
The before cited nausea-related stimuli include those from plant-derived irritants, bacterial toxins, viral proteins plus other classic emetic stimuli originated in the gastrointestinal tract are first identify by the vagal afferent pathway responding from gastrointestinal mucosa barrier compose by specialized sensory cells and epithelial cells, including enterochromaffin cells and enteroendocrine cells which detect chemical cues such as irritants/nutrients which release hormones and neurotransmitters, like GLP-1, serotonin, cholecystokinin, and substance P receptor, cysteinyl leukotriene receptors, and interleukin receptors [43-47].
Blocking substance P receptors and serotonin receptors is the main pharmacological role of most anti-nausea drugs that act on the sensory vagal neurons (NTS) and other brainstem regions (AP), among other targets.
Brief comments on GLP1R agonists
Glucagon-Like Peptide-1 (GLP-1) receptor agonists medication. It’s quite common (60%) associated to nausea and vomiting but its highly involved in the mechanism to reduce level of sugar via pancreatic control and blocking of feeding primarily originates from targets in the hypothalamic nucleus and brainstem mainly in areas outside the BBB and close to circumventricular organs in the brain facilitating an easy access of the GLP1R agonist to them [48-50].
Therefore, we hypothesized that the AP could trigger nausea and vomiting in response to GLP1R agonist intake. At the same time, appetite suppression is associated with the inhibitory activity of GLP1R+ neurons in the paraventricular hypothalamus or with increased catabolic activity. On the other hand, when GLP1R+ neurons in the AP and NTS are damaged, the GLP1R agonist-induced feeding suppression disappears. Based on the previous postulate, we hypothesize that the GLP1R+ neuronal population in the brainstem is prominently involved in the weight-loss effects of GLP1R agonists. However, we do not have sufficient evidence to determine whether the major source of the GLP-1 neuropeptide is the brainstem glucagon neurons or the enteric autonomic neurons, as reported by other authors [51].
GLP1R+ neurons located at the dorsomedial region of the hypothalamus can modulate preingestive satiation [54].
Brief comments on metabolism
The relationship between the AP neural pathway for regulation of metabolism (metabolic syndrome), wasting syndrome, nausea and cancer cachexia mediated by IL-6 expression on the AP neurons leading to intractable anorexia as has documented by many investigators [53].
Other authors have documented that AP GFRAL (+) neurons can induce ketogenesis, hypothermia, and lipid oxidation [54], and we speculate that, through the sympathetic/parasympathetic pathway, AP neurons can exert a remarkable influence/control over metabolism.
On top of that, AP also plays a key role in controlling energy, which influenced by influences, modulating agonists, and modulating cancer-related weight previously cited as cachexia.
Notwithstanding, there are aspects related to AP interrogation that we cannot answer due to a lack of evidence found in our review, such as the exact mechanism of nausea and vomiting in cases under chemotherapy, affected by food and poisoning/allergy, and pregnancy. We do not know whether any additional signalling elements beyond IL-6 and GDF15 involved in the pathophysiology of nausea. We do not know the exact biochemical influence of the vagus nerve on the nausea response.
Brief comments on the drug therapy algorithm for Area Postrema Syndrome (APS)
APS is an uncommon clinical condition characterized by intractable nausea and vomiting in response to direct AP injury. Recently, Stoica et al. reviewed the medical literature seeking novel management of refractory central nausea and vomiting, highlighting other issues apart from targeting conventional dopaminergic/serotonergic/ histaminergic blockade at central centres of hyperemesis and proposing that the disruption of the baseline inhibitory tone of AP signals is the main source of APS [55].
The vomiting reflex modulated by the Dorsal Vagal Complex (DVC), which is primarily composed of the Nucleus Tractus Solitarius (NTS) and the AP [56].
There are few published investigations in the medical literature on the treatment of the central causes of intractable vomiting. Therefore, medical doctors faced with challenging decisions when presented with cases whose nausea and emesis respond weakly to first-line antiemetic drugs.
Because centrally induced emesis has a different pathogenesis compared with chemotherapy-related nausea/vomiting, the management of both has remarkable differences. In contrast, APS may lead an inhibitory tone on the feeling of nausea and an efferent motor reflex on emesis, which may be reinforced by afferent gastrointestinal signals or neural input from chemoreceptors stimulated via chemicals in the serum. It has documented that direct injury of the AP leads to a hyperemetic state, and this situation is associated with improper failure to respond to the first line of causative drugs caused by inappropriate blockage of long-lasting neurons belonging to the vomiting centre driven by serotonin/substance P [57].
Specific brain areas send inputs to the DVC, including the insula cortex, vestibular nucleus, enteric vagal afferents, and circulating molecules [58-61].
The emetic response after receiving the internal/external stimuli (e.g., bacteria, cardiac ischemia, and toxins) executed by the dorsal motor nucleus of the vagal nerve (DMX).
The major therapeutic challenge in patients presenting APS is to control intractable nausea and vomiting, which leads to patients’ discomfort, lack of satisfaction/safety plus metabolic and electrolyte complications, including cachexia and vitamin deficiencies. There are ways to modulate the AP’s function. We hypothesized, that the best one is to administer medical drugs able to produce local release of serotonin, which then binds to enteric vagal afferents to send a corresponding signal to the AP and NTS by stimulation of the enterochromaffin cells and to modulate circulating toxins causes Substance P (SP)- mediated initiation to control the emetic response as has been proposed in cases with chemotherapy-induced nausea and vomiting [62]. Based on the pharmacological action of strong antiemetic drugs able to target those receptors located at the AP, such as Neurokinin-1 (NK- 1) SP receptor and type-3 serotonin receptor (5HT-3) [63].
Another way to control those refractory manifestations is to modulate receptors widely distributed in the dorsal vagal complex, such as Calcitonin Gene-Related Peptide (CGRP) receptors, histaminergic (H1) receptors, muscarinic receptors, Dopamine type 2 (D2) receptors, the endocannabinoid system, and gastric peptide hormone receptors [64].
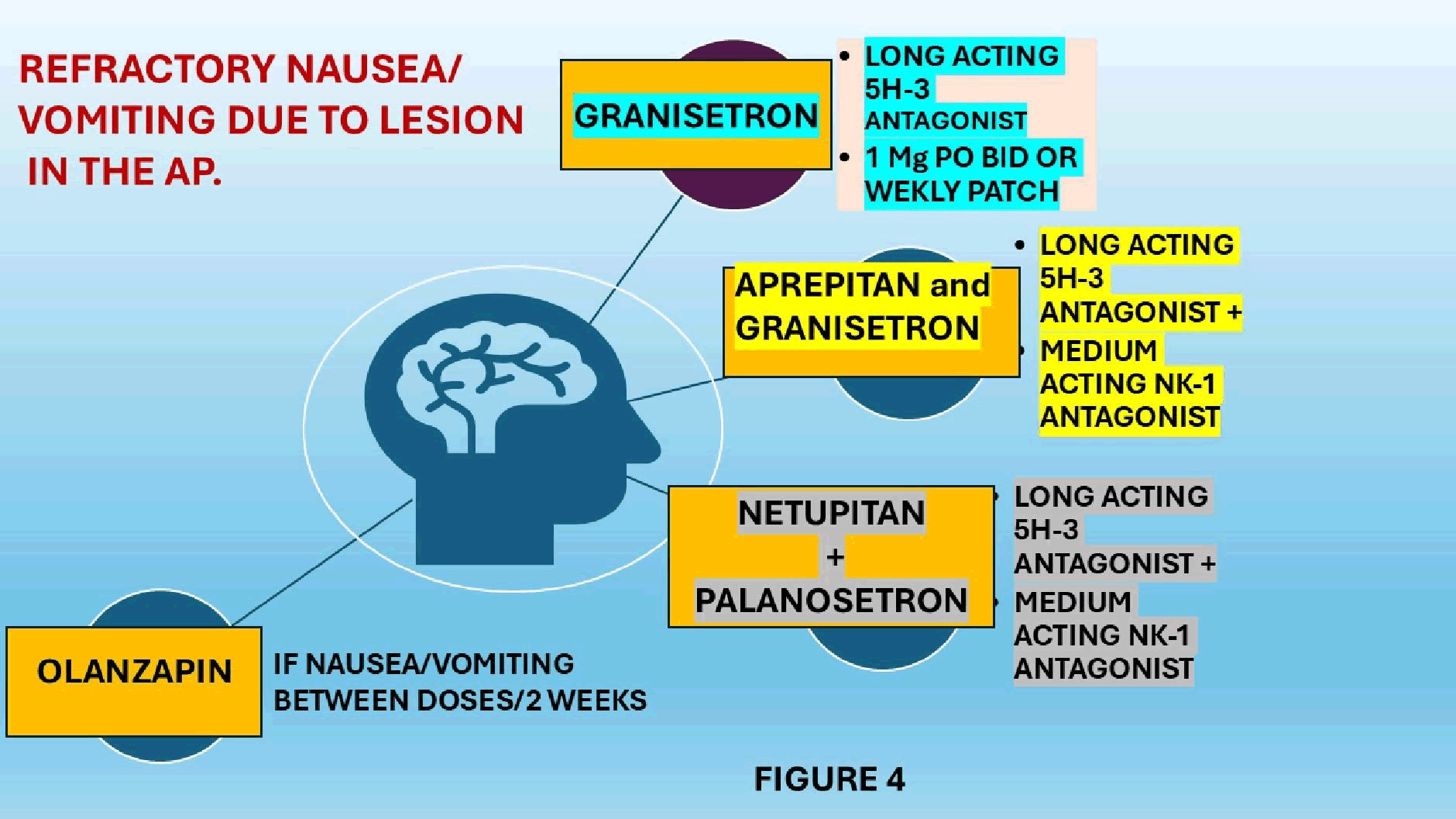
As previously cited, the main cause of the APS leading to intractable nausea and vomiting is in the brainstem, and its management includes proper correction of electrolyte/ nutritional disturbances and rehydration, plus first-line GNT drugs with anti-D2/D3 activity such as prochlorperazine, promethazine, ondansetron, metoclopramide, or anti-5HT-3. We personally recommended a combination of anti-dopaminergic medications and anti-5HT-3, and if the patient does not respond, then we suggest administering olanzapine with highly strict observation. In patients not on chemotherapy, the best choice is netupitant and palonosetron [65]. This longer-acting antiemetic targets 5-HT3 and NK1 receptors, with careful assessment of their interaction to avoid toxicity and other complications, including QT interval prolongation and other cardiac problems. In contrast, APS cases with refractory nausea and vomiting, or other combinations of NK-1 receptor antagonist/5-HT3 blockers and D2 blockers, continue to be a good decision. Another choice is administering Granisetron orally twice a day or even in a patch as a weekly therapy.
New evidence from molecular investigations has revealed remarkable therapeutic targets, such as the Transient Receptor Potential Vanilloid-1 (TRPV1), Gastric Inhibitory Polypeptide Receptor (GIPR) and Glucagon-Like Peptide-1 (GLP-1) receptors, which are expressed in the AP and have the capacity to modulate nausea and vomiting separately by acting as chemical triggers.
On the other hand, other investigators recommend prescribing netupitant and palonosetron to control refractory nausea and vomiting due to AP lesions based on their results (Figure 4) [66].
Figure 4: List of the best therapeutic drugs recommended to control refractory nausea and vomiting secondary to APS
It is important to highlight the need to provide safe access to medical care for patients taking the netupitant and palonosetron combination tablets. The unwelcome news is that combination tables are quite expensive, but the good news is that they only need to prescribe weekly, and they have a net cost-saving effect [67].
Conclusion
Only eight studies reported novel therapeutic procedures to control refractory nausea and vomiting due to AP disorder, that we discussed in this article. Based on bibliography investigation done, the best therapeutic drugs to control refractory nausea and vomiting due to APS are: Granisetron, Aprepitant, Netupitant, Palonosetron and Olanzapine prescribing alone or in combination according to the clinical respond and under safe supervision.
Acknowledgment
To thanks to Prof Thozama Dubula for his support.
Ethics Statement
This review does not require ethical approval.
Conflicts of Interest
Authors of this review report there is not conflicts of interest.
References
- R.M. Navari, M. Aapro, Antiemetic prophylaxis for chemotherapyâinduced nausea and vomiting, N Engl J Med, 374(2016):1356–1367.
[Crossref] [Google Scholar] [PubMed]
- M.J. McKinley, D.A. Denton, P.J. Ryan, S.T. Yao, A. Stefanidis, et al. From sensory circumventricular organs to cerebral cortex: Neural pathways controlling thirst and hunger, J Neuroendocrinol, 31(2019):e12689.
[Crossref] [Google Scholar] [PubMed]
- K. Scorza, A. Williams, J.D. Phillips, J. Shaw, Evaluation of nausea and vomiting, Am Fam Physician, 76(2007):76–84.
[Google Scholar] [PubMed]
- P.M. Gross, K.M. Wall, D.S. Wainman, S.W. Shaver, Subregional topography of capillaries in the dorsal vagal complex of rats: II. Physiological properties, J Comp Neurol, 306(1991):83–94.
[Crossref] [Google Scholar] [PubMed]
- K. Bruce, A.N. Garrido, S.Y. Zhang, T.K.T. Lam, Regulation of energy and glucose Homeostasis by the nucleus of the solitary tract and the area postrema, Endocrinol Metab (Seoul), 39(2024):559–568.
[Crossref] [Google Scholar] [PubMed]
- S.Y. Zhang, K. Bruce, Z. Danaei, R.J.W. Li, D.R. Barros, et al. Metformin triggers a kidney GDF15âdependent area postrema axis to regulate food intake and body weight, Cell Metab, 35(2023):875–886.
[Crossref] [Google Scholar] [PubMed]
- D.A. Prentice, R. Ambati, L.K. Kho, T. Jenkins, P.M. Parizel, Seronegative autoimmune encephalomyelitis with area postrema symptoms, Case Rep Neurol, 17(2025):50–56.
[Crossref] [Google Scholar] [PubMed]
- J.R. MuñozâZúñiga, A.M. TamayoâDelgado, A.M. ShinchiâTanaka, J.C. Márquez, A. EcheverriâGarcía, et al. Postrema area syndrome in the context of systemic lupus erythematosus: Case report, Heliyon, 11(2024):e40614.
[Crossref] [Google Scholar] [PubMed]
- P. Stancu, N. Sanda, K.O. Lovblad, N. Guinand, A. Kleinschmidt, et al. Area postrema syndrome as the only sign of medullary infarction adjacent to area postrema, eNeurologicalSci, 39(2025):100563.
[Crossref] [Google Scholar] [PubMed]
- C. Courtney, C. Franconi, F. Dharsono, D. Prentice, Giant cell arteritis manifesting as area postrema syndrome: A case report, Cureus, 17(2025):e93470.
[Crossref] [Google Scholar] [PubMed]
- Q. Li, J. Teng, Area postrema syndrome in patients with autoimmune glial fibrillary acidic protein astrocytopathy, Front Neurol, 16(2025):1538602.
[Crossref] [Google Scholar] [PubMed]
- S.W. Shaver, J.J. Pang, K.M. Wall, N.M. Sposito, P.M. Gross, Subregional topography of capillaries in the dorsal vagal complex of rats: I. Morphometric properties, J Comp Neurol, 306(1991):73–82.
[Crossref] [Google Scholar] [PubMed]
- I.L. Bernstein, M. Chavez, D. Allen, E.M. Taylor, Area postrema mediation of physiological and behavioral effects of lithium chloride in the rat, Brain Res, 575(1992):132–137.
[Crossref] [Google Scholar] [PubMed]
- E. Shosha, D. Dubey, J. Palace, I. Nakashima, A. Jacob, et al. Area postrema syndrome: Frequency, criteria, and severity in AQP4âIgGâpositive NMOSD, Neurology, 91(2018):1642–1651.
[Crossref] [Google Scholar] [PubMed]
- L.F.I. Valdes, H. FoyacaâSibat, Impact of gut dysbiosis on neuromyelitis optica spectrum disorder: Implications for alcohol and substance use, J Drug Alcohol Res, 13(2024):236418.
- J. LaraâGarcía, J. RomoâMartínez, J.J. DeâLaâCruzâCisneros, M.A. OlveraâOlvera, L.J. MárquezâBejarano, Unmasking the area postrema on MRI: Utility of 3D FLAIR, 3DâT2, and 3DâDIR sequences in a case–control study, J Imaging, 11(2025):16.
[Crossref] [Google Scholar] [PubMed]
- T.F.S. Des, A concise historical perspective of the area postrema structure and function, Arq Neuropsiquiatr, 78(2020):121–123.
- C. Zhang, Neural pathways of nausea and roles in energy balance, Curr Opin Neurobiol, 90(2025):102963.
[Crossref] [Google Scholar] [PubMed]
- H.L. Borison, Area postrema: Chemoreceptor circumventricular organ of the medulla oblongata, Prog Neurobiol, 32(1989):351–390.
[Crossref] [Google Scholar] [PubMed]
- C.C. Horn, Why is the neurobiology of nausea and vomiting so important?, Appetite, 50(2008):430–434.
[Crossref] [Google Scholar] [PubMed]
- P.L. Andrews, Physiology of nausea and vomiting, Br J Anaesth, 69(1992):2S–19S.
[Crossref] [Google Scholar] [PubMed]
- G.J. Sanger, P.L. Andrews, Treatment of nausea and vomiting: Gaps in our knowledge, Auton Neurosci, 129(2006):3–16.
[Crossref] [Google Scholar] [PubMed]
- H. Yin, Y. Wang, Y. Xu, Neuromyelitis optica spectrum disorders with recurrent syncope due to area postrema syndrome: A case report and literature review, BMC Neurol, 25(2025):339.
[Crossref] [Google Scholar] [PubMed]
- H.L. Borison, Area postrema: Chemoreceptor circumventricular organ of the medulla oblongata, Prog Neurobiol, 32(1989):351–390.
[Crossref] [Google Scholar] [PubMed]
- C.J. Price, T.D. Hoyda, A.V. Ferguson, The area postrema: A brain monitor and integrator of systemic autonomic state, Neuroscientist, 14(2008):182–194.
[Crossref] [Google Scholar] [PubMed]
- A.D. Miller, R.A. Leslie, The area postrema and vomiting, Front Neuroendocrinol, 15(1994):301–332.
[Crossref] [Google Scholar] [PubMed]
- J.Y. Hsu, S. Crawley, M. Chen, D.A. Ayupova, D.A. Lindhout, et al. Nonâhomeostatic body weight regulation through a brainstemârestricted receptor for GDF15, Nature, 550(2017):255–259.
[Crossref] [Google Scholar] [PubMed]
- R.D. Palmiter, The parabrachial nucleus: CGRP neurons function as a general alarm, Trends Neurosci, 41(2018):280–293.
[Crossref] [Google Scholar] [PubMed]
- M.C. Chiang, A. Bowen, L.A. Schier, D. Tupone, O. Uddin, et al. Parabrachial complex: a hub for pain and aversion, J Neurosci, 39(2019):8225–8230.
[Crossref] [Google Scholar] [PubMed]
- J.L. Pauli, J.Y. Chen, M.L. Basiri, S. Park, M.E. Carter, et al. Molecular and anatomical characterization of parabrachial neurons and their axonal projections, Elife, 11(2022).
[Crossref] [Google Scholar] [PubMed]
- C. Zhang, J.A. Kaye, Z. Cai, Y. Wang, S.L. Prescott, et al. Area postrema cell types that mediate nauseaâassociated behaviours, Neuron, 109(2021):461–472.
[Crossref] [Google Scholar] [PubMed]
- S. Han, M.T. Soleiman, M.E. Soden, L.S. Zweifel, R.D. Palmiter, Elucidating an affective pain circuit that creates a threat memory, Cell, 162(2015):363–374.
[Crossref] [Google Scholar] [PubMed]
- J.Y. Chen, C.A. Campos, B.C. Jarvie, R.D. Palmiter, Parabrachial CGRP neurons establish and sustain aversive taste memories, Neuron, 100(2018):891–899.e895.
[Crossref] [Google Scholar] [PubMed]
- C.A. Zimmerman, S.S. Bolkan, A. PanâVazquez, B. Wu, E.F. Keppler, et al. A neural mechanism for learning from delayed postingestive feedback, bioRxiv, (2024).
- A. Costa, M. Ai, N. Nunn, I. Culotta, J. Hunter, et al. Anorectic and aversive effects of GLPâ1 receptor agonism are mediated by brainstem cholecystokinin neurons and modulated by GIP receptor activation, Mol Metab, 55(2022):101407.
[Crossref] [Google Scholar] [PubMed]
- R.J. Samms, R. Cosgrove, B.M. Snider, E.C. Furber, B.A. Droz, et al. GIPR agonism inhibits PYYâinduced nauseaâlike behaviour, Diabetes, 71(2022):1410–1423.
[Crossref] [Google Scholar] [PubMed]
- A. Mollet, S. Gilg, T. Riediger, T.A. Lutz, Infusion of the amylin antagonist AC 187 into the area postrema increases food intake in rats, Physiol Behav, 81(2004):149–155.
[Crossref] [Google Scholar] [PubMed]
- A. Cabral, M.P. Cornejo, G. Fernandez, P.N. De Francesco, G. GarciaâRomero, et al. Circulating ghrelin acts on GABA neurons of the area postrema and mediates gastric emptying in male mice, Endocrinology, 158(2017):1436–1449.
[Crossref] [Google Scholar] [PubMed]
- Q. Sun, D. van de Lisdonk, M. Ferrer, B. Gegenhuber, M. Wu, et al. Area postrema neurons mediate interleukinâ6 function in cancer cachexia, Nat Commun, 15(2024):4682.
[Crossref] [Google Scholar] [PubMed]
- F. Jagot, R. GastonâBreton, A.J. Choi, M. Pascal, L. Bourhy, et al. The parabrachial nucleus elicits a vigorous corticosterone feedback response to the proâinflammatory cytokine ILâ1beta, Neuron, 111(2023):2367–2382.e2366.
[Crossref] [Google Scholar] [PubMed]
- L. Bai, S. Mesgarzadeh, K.S. Ramesh, E.L. Huey, Y. Liu, et al. Genetic identification of vagal sensory neurons that control feeding, Cell, 179(2019):1129–1143.e1123.
[Crossref] [Google Scholar] [PubMed]
- Q. Zhao, C.D. Yu, R. Wang, Q.J. Xu, R. Dai Pra, et al. A multidimensional coding architecture of the vagal interoceptive system, Nature, 603(2022):878–884.
[Crossref] [Google Scholar] [PubMed]
- H.E. Tan, A.C. Sisti, H. Jin, M. Vignovich, M. Villavicencio, et al. The gutâbrain axis mediates sugar preference, Nature, 580(2020):511–516.
[Crossref] [Google Scholar] [PubMed]
- M. Li, H.E. Tan, Z. Lu, K.S. Tsang, A.J. Chung, et al. Gutâbrain circuits for fat preference, Nature, 610(2022):722–730.
[Crossref] [Google Scholar] [PubMed]
- M. Hayashi, J.A. Kaye, E.R. Douglas, N.R. Joshi, F.M. Gribble, et al. Enteroendocrine cell lineages that differentially control feeding and gut motility, Elife, 12(2023).
[Crossref] [Google Scholar] [PubMed]
- K.K. Touhara, N.D. Rossen, F. Deng, T. Chu, A.M. Harrington, et al. Crypt and villus enterochromaffin cells are distinct stress sensors in the gut, bioRxiv, (2024).
- M. Hayashi, J.A. Kaye, E.R. Douglas, N.R. Joshi, F.M. Gribble, et al. Enteroendocrine cell lineages that differentially control feeding and gut motility, Elife, 12(2023).
[Crossref] [Google Scholar] [PubMed]
- J.M. Adams, H. Pei, D.A. Sandoval, R.J. Seeley, R.B. Chang, et al. Liraglutide modulates appetite and body weight through glucagonâlike peptide 1 receptorâexpressing glutamatergic neurons, Diabetes, 67(2018):1538–1548.
[Crossref] [Google Scholar] [PubMed]
- S. Gabery, C.G. Salinas, S.J. Paulsen, J. AhnfeltâRonne, T. Alanentalo, et al. Semaglutide lowers body weight in rodents via distributed neural pathways, JCI Insight, 5(2020).
[Crossref] [Google Scholar] [PubMed]
- A.N. Webster, J.J. Becker, C. Li, D.C. Schwalbe, D. Kerspern, et al. Molecular connectomics reveals a glucagonâlike peptide 1 sensitive neural circuit for satiety, bioRxiv, (2024).
[Crossref] [Google Scholar] [PubMed]
- D.I. Brierley, M.K. Holt, A. Singh, A. de Araujo, M. McDougle, et al. Central and peripheral GLPâ1 systems independently suppress eating, Nat Metab, 3(2021):258–273.
[Crossref] [Google Scholar] [PubMed]
- K.S. Kim, J.S. Park, E. Hwang, M.J. Park, H.Y. Shin, et al. GLPâ1 increases preingestive satiation via hypothalamic circuits in mice and humans, Science, 385(2024):438–446.
[Crossref] [Google Scholar] [PubMed]
- A.P. Coll, M. Chen, P. Taskar, D. Rimmington, S. Patel, et al. GDF15 mediates the effects of metformin on body weight and energy balance, Nature, 578(2020):444–448.
- L. Engstrom Ruud, F. FontâGirones, J. Zajdel, L. Kern, J. TeixidorâDeulofeu, et al. Activation of GFRAL (+) neurons induces hypothermia and glucoregulatory responses associated with nausea and torpor, Cell Rep, 43(2024).
[Crossref] [Google Scholar] [PubMed]
- S. Stoica, C. Hogge, B.J. Theeler, Refractory nausea and vomiting due to central nervous system injury: A focused review, Life (Basel), 15(2025):1021.
[Crossref] [Google Scholar] [PubMed]
- W. Zhong, O. Shahbaz, G. Teskey, A. Beever, N. Kachour, et al. Mechanisms of nausea and vomiting: Current knowledge and recent advances in intracellular emetic signaling systems, Int J Mol Sci, 22(2021):5797.
[Crossref] [Google Scholar] [PubMed]
- C. Hang, L.K. Vincelette, F. Reimann, S.D. Liberles, A brainstem circuit for nausea suppression, Cell Rep, 39(2022):110953.
[Crossref] [Google Scholar] [PubMed]
- C. Stephani, G. FernandezâBaca Vaca, R. Maciunas, M. Koubeissi, H.O. Lüders, Functional neuroanatomy of the insular lobe, Brain Struct Funct, 216(2011):137–149.
- I. Indovina, R. Riccelli, G. Chiarella, C. Petrolo, A. Augimeri, et al. Role of the insula and vestibular system in patients with chronic subjective dizziness: An fMRI study using soundâevoked vestibular stimulation, Front Behav Neurosci, 9(2015):334.
- E.E. Benarroch, Insular cortex: Functional complexity and clinical correlations, Neurology, 93(2019):932–938.
[Crossref] [Google Scholar] [PubMed]
- S. Cauzzo, K. Singh, M. Stauder, M.G. GarcíaâGomar, N. Vanello, et al. Functional connectome of brainstem nuclei involved in autonomic, limbic, pain and sensory processing in living humans from 7 Tesla resting state fMRI, Neuroimage, 250(2022):118925.
- P.J. Hesketh, Chemotherapyâinduced nausea and vomiting, N Engl J Med, 358(2008):2482–2494.
[Crossref] [Google Scholar] [PubMed]
- C. Rojas, M. Raje, T. Tsukamoto, B.S. Slusher, Molecular mechanisms of 5âHT(3) and NK(1) receptor antagonists in prevention of emesis, Eur J Pharmacol, 722(2014):26–37.
[Crossref] [Google Scholar] [PubMed]
- R.J. Wickham, Revisiting the physiology of nausea and vomitingâchallenging the paradigm, Support Care Cancer, 28(2019):13–21.
[Crossref] [Google Scholar] [PubMed]
- C. Zhang, J.A. Kaye, Z. Cai, Y. Wang, S.L. Prescott, et al. Area postrema cell types that mediate nauseaâassociated behaviors, Neuron, 109(2021):472.
[Crossref] [Google Scholar] [PubMed]
- M. Aapro, K. Jordan, F. Scotté, L. Celio, M. Karthaus, et al. Netupitantâpalonosetron (NEPA) for preventing chemotherapyâinduced nausea and vomiting: From clinical trials to daily practice, Curr Cancer Drug Targets, 22(2022):806–824.
[Crossref] [Google Scholar] [PubMed]
- S.H. Park, G. Binder, S. Corman, M. Botteman, Budget impact of netupitant/palonosetron for the prevention of chemotherapyâinduced nausea and vomiting, J Med Econ, 22(2019):840–847.
[Crossref] [Google Scholar] [PubMed]
Copyright: © 2026 Lourdes de Fatima Ibanez Valdes, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.